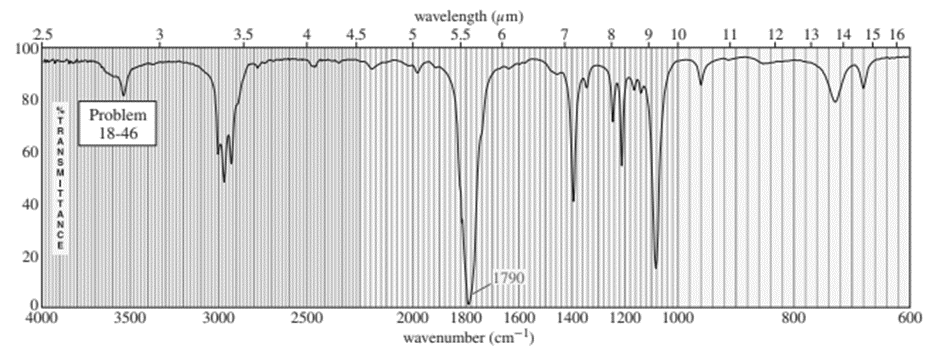
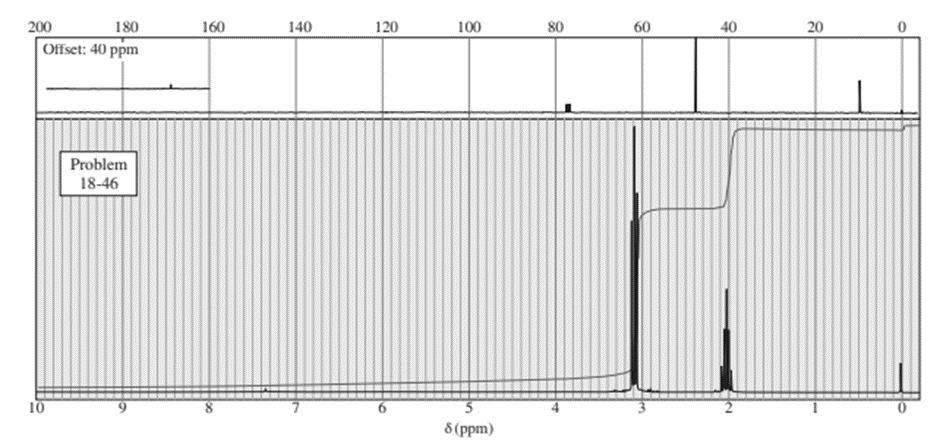
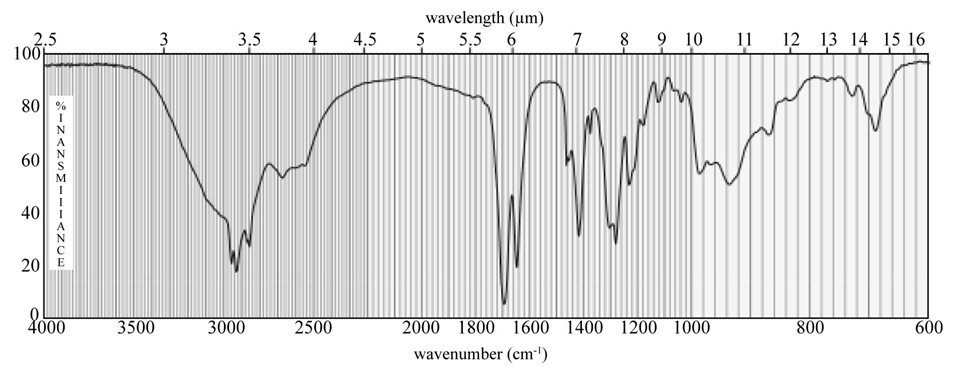
Chapter 20: Q-6-8P (page 1038)
(a) Propose a mechanism for the following reaction: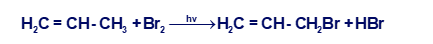
(b) Use the bond-dissociation enthalpies given in Table 4-2 (page 203) to calculate the value of ∆H° for each step shown in your mechanism. (The BDE for is about 280 kJ/mol, or 67 kcal/mol.) Calculate the overall value of ∆H° for the reaction. Are these values consistent with a rapid free-radical chain reaction?
Short Answer
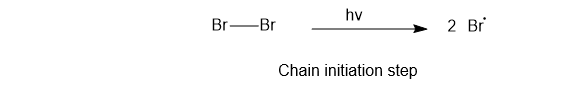
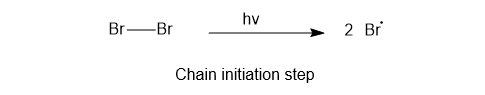
1) Chain initiation
In this step bromine free radical is formed.
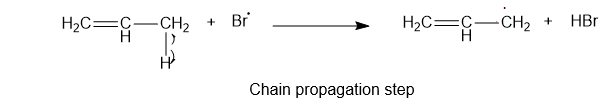
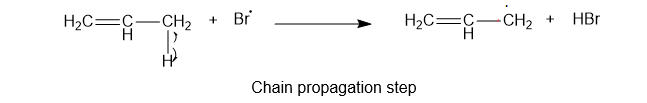
2) The second step is the chain propagation step. Thebrmonie free radical reacts with the hydrogen atom of an allylic carbon atom.
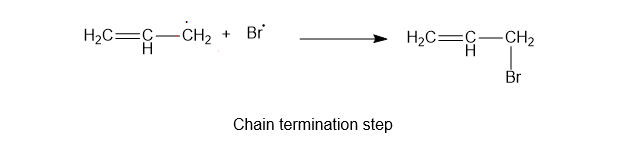
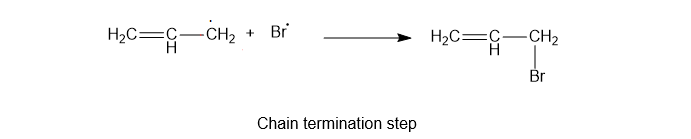
3) Chain termination reaction
The required product, the alkyl halide, will be formed in this.