Chapter 20: Q36P (page 1074)
Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
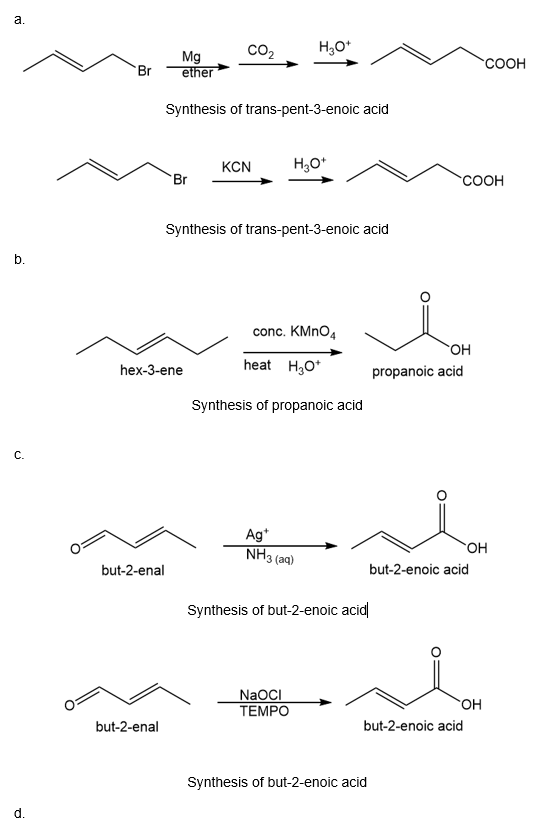
(a)trans-1-�����dz��Dz��ܳ�-2-��Ա�→trans-pent-3-enoic acid (two ways)
(b)hex-3-ene→propanoic acid
(c) but-2-enal→but-2-enoic acid
(d) hexanoic acid→hexanal
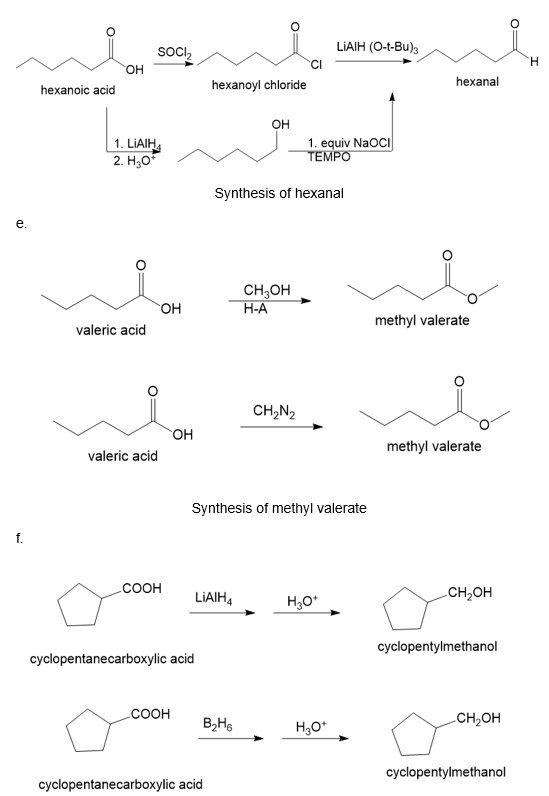
(e)
(f)
(g)
(h)
Short Answer
Synthesis of cyclopentylmethanol
Step by step solution
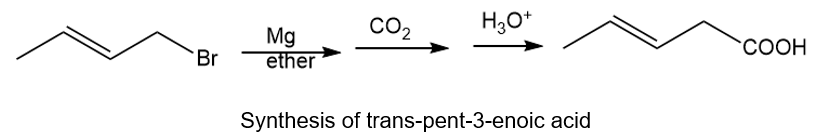
About trans-1-bromobut-2-ene
(a) The organic compound that bears four carbons in the parent chain and a double bond at the second carbon with trans configuration is trans-1-bromobut-2-ene.
The trans configuration is for the higher atomic groups on the opposite side of the double bond.
About trans-pent-3-oic acid
The organic compound that bears five carbons in the parent chain and a COOH at the third carbon with trans configuration is trans-pent-3-oic acid. The trans configuration is for the higher atomic groups on the opposite side of the double bond.
About the synthesis
The given transformation can be carried out by treating trans-1-bromobut-2-ene with magnesium in the presence of ether followed by treating with carbon dioxide and then treating with acid gives the desired product. The reaction is given below.
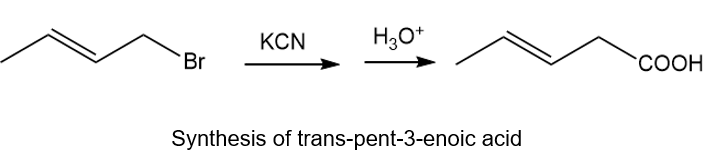
About the synthesis
Another method is by treating trans-1-bromobut-2-ene with KCN followed by treating with acid will give the desired product. The reaction is shown below.
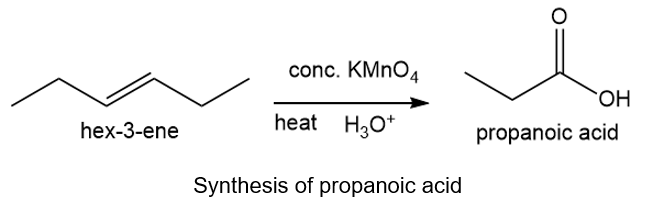
About hex-3-ene and propanoic acid
(b) The organic compound with six carbons in the parent chain and one double bond at the third carbon is hex-3-ene. The propanoic acid bears three carbons in the parent chain and one carboxylic acid group.
About the reaction
The conversion of hex-3-ene to propanoic acid can be carried out by heating hex-3-ene with concentrated potassium permanganate in the presence of acid. The reaction is shown below.
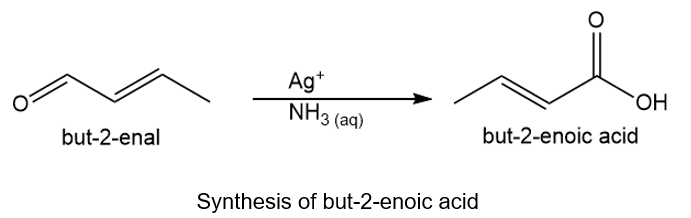
About but-2-enal and but-2-enoic acid
(c) The organic compound with four carbons in the parent chain and one aldehyde group at the second carbon is but-2-enal. The but-2-enoic acid bears four carbons in the parent chain and one carboxylic acid group at the second carbon.
About the reaction
The conversion of but-2-enal to but-2-enoic acid can be carried out by treating but-2-enal with silver ions in the presence of aqueous ammonia. The reaction is shown below.
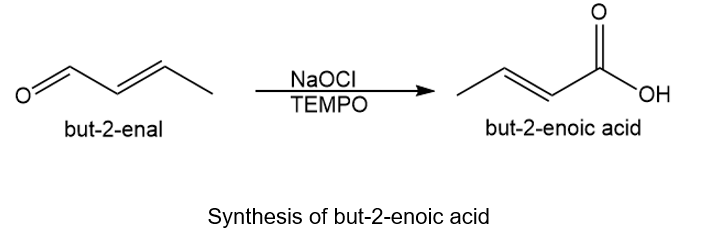
About the reaction
Another method is by treating but-2-enal with NaOCl. The reaction is shown below.
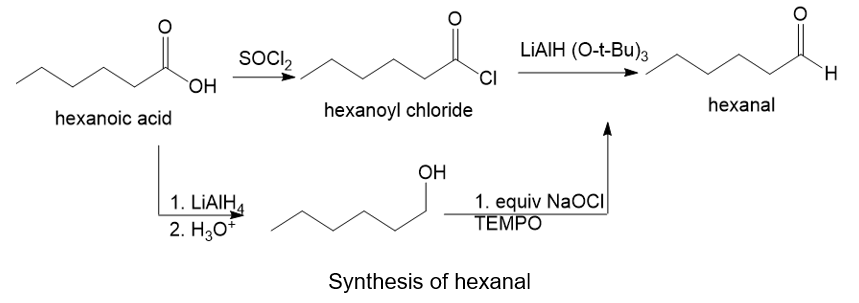
About hexanoic acid and hexanal
(d) The organic compound with six carbons in the parent chain and one carboxylic acid group at the first carbon is hexanoic acid. The hexanal bears four carbons in the parent chain and one aldehyde group at the first carbon.
About the reaction
The conversion of hexanoic acid to hexanal can be carried out by treating hexanoic acid with thionyl chloride followed by the addition of lithium hydroxide.
Another method is by treating hexanoic acid with lithium aluminium hydride and acid followed by treating with NaOCl gives hexanal. The reaction is shown below.
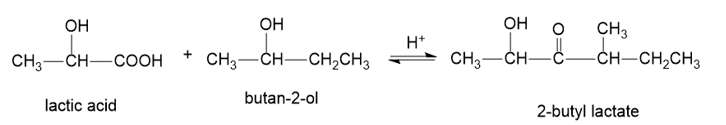
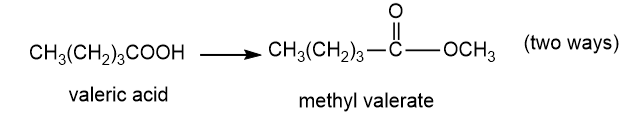
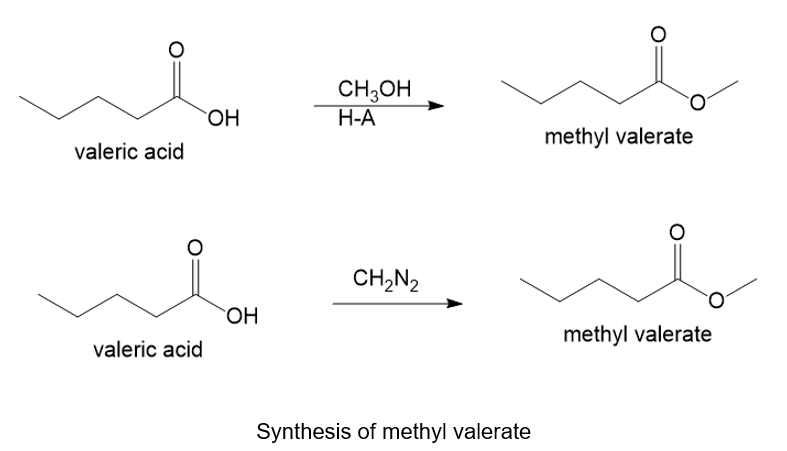
About valeric acid
(e) The organic compound with five carbons in the parent chain and one carboxylic acid group at the first carbon is valeric acid. This compound is also known as pentanoic acid.
About the reaction
The conversion of valeric acid to methyl valerate can be carried out by treating valeric acid with methanol in the presence of an acid. Another method is by treating valeric acid with diazomethane gives methyl valerate. The reaction is shown below.
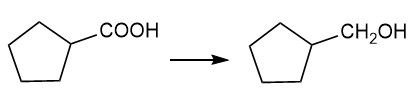
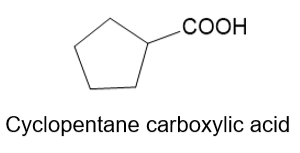
About cyclopentane carboxylic acid
(f) The organic compound with five carbons in the cyclic parent chain and one carboxylic acid group at the first carbon is cyclopentane carboxylic acid. This compound has the following structure.
About the reaction
The conversion of cyclopentane carboxylic acid to cyclopentylmethanol can be carried out by treating cyclopentane carboxylic acid with lithium aluminium hydride followed by treatment with acid.
Another method is by treating cyclopentane carboxylic acid with diborane in the presence of acid gives cyclopentylmethanol. The reaction is shown below.
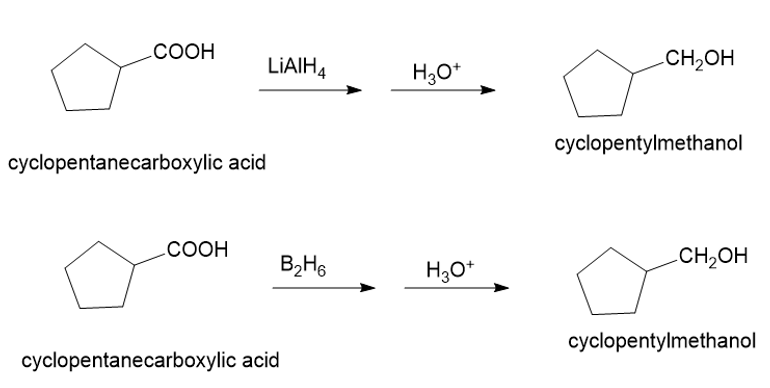
Synthesis of cyclopentylmethanol
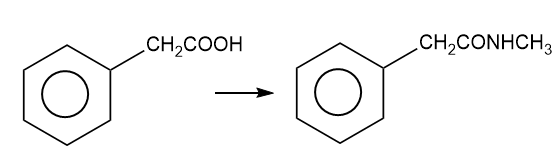
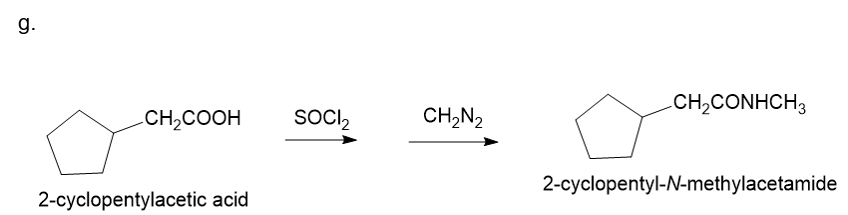
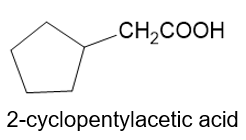
About 2-cycclopentyl acetic acid
(g) The organic compound with acetic acid containing cyclopentyl ring at the second carbon is 2-cyclopentyl acetic acid. This compound has the following structure.
About the reaction
The conversion of 2-cyclopentylacetic acid to 2-cyclopentyl-N-methylacetamide can be carried out by treating 2-cyclopentylacetic acid with thionyl chloride followed by treatment with diazomethane. The reaction is shown below.
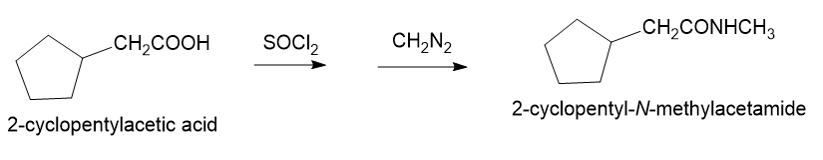
Synthesis of 2-cyclopentyl-N-methylacetamide
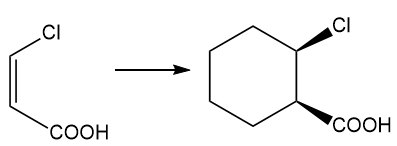
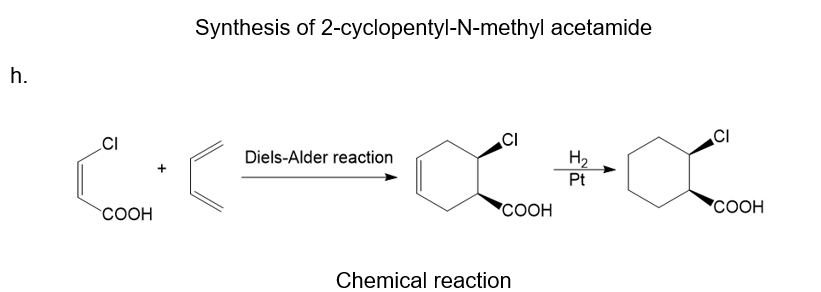
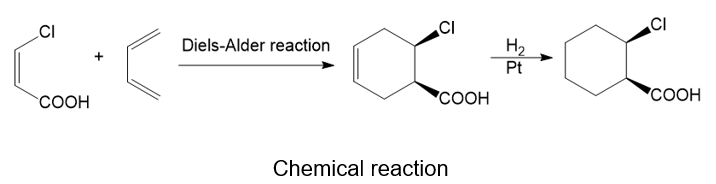
About the Diels-Alder reaction
(h) The chemical reaction that occurs between a substituted alkene and conjugated diene is the Diels-Alder reaction. This reaction is related to a pericyclic reaction.
About the reaction
The given reaction can be carried out by the Diels-Alder reaction which is followed by treatment with hydrogen in the presence of a platinum catalyst. The reaction is shown below.

Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their learning with 91Ӱ��!