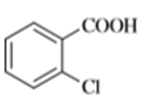
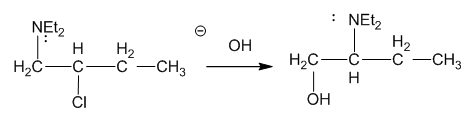
Chapter 20: Q9P. (page 1055)
Question: Draw all four resonance forms of the fragment at m/z 73 in the mass spectrum of pentanoic acid.
Short Answer
Answer
The four resonance forms of the fragment at m/z 73 in pentanoic acid are shown:
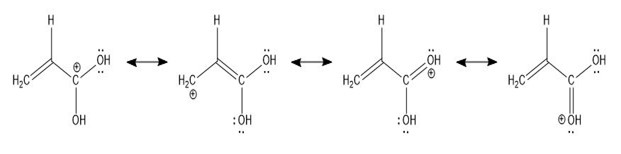
Four resonance forms of pentanoic acid have fragments at m/z 73