Chapter 20: Q14P c. (page 1062)
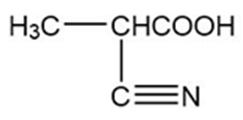
Question: A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.
- Draw the resonance forms of a carboxylic acid that is protonated on the hydroxy oxygen atom.
- Compare the resonance forms with those given previously for an acid protonated on the carbonyl oxygen atom.
- Explain why the carbonyl oxygen atom of a carboxylic acid is more basic than the hydroxy oxygen.
Short Answer
Answer
(c) The intermediates formed in the protonation of carbonyl oxygen are more stable than the intermediate obtained from the protonation of the hydroxyl oxygen atom.


 ,
, ,
,