Chapter 30: Question 30.32 (page 1225)
Draw the structure of the polymer that results from anionic polymerization of p-trichloromethylstyrene using ethylene oxide as the electrophile to terminate the chain.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 30: Question 30.32 (page 1225)
Draw the structure of the polymer that results from anionic polymerization of p-trichloromethylstyrene using ethylene oxide as the electrophile to terminate the chain.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
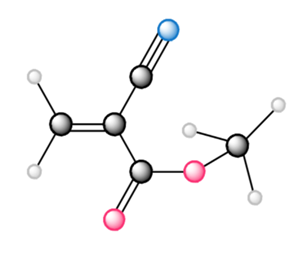
Draw the structure of the polymer formed by chain-growth polymerization of each monomer.
a.
b.
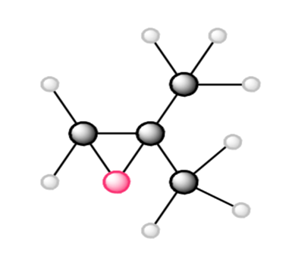
What monomer(s) are used to prepare each polymer or copolymer?
a.
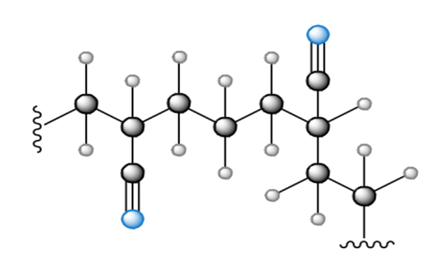
b.
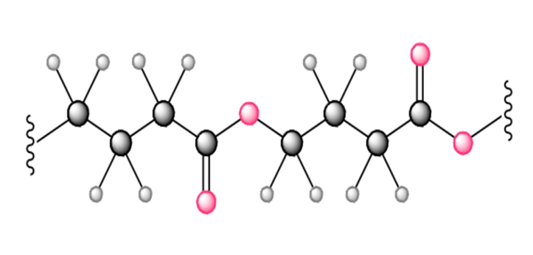
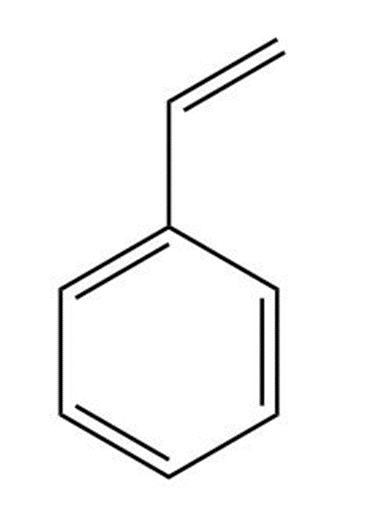
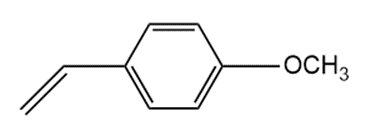
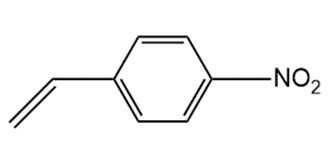
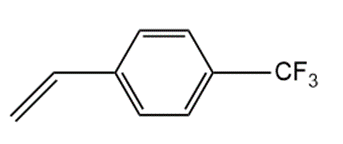
Although styrene undergoes both cationic and anionic polymerization equally well, one method is often preferred with substituted styrenes. Which method is preferred with each compound? Explain.
a.
b.
c.
d.
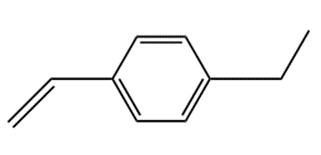
The reaction of urea and formaldehyde role="math" localid="1648635328169" forms a highly cross-linked polymer used in foams. Suggest a structure for this polymer. [Hint: Examine the structures of Bakelite (Figure 30.7) and Melmac (Problem 30.58).]
What polymers are formed from each monomer?
a.
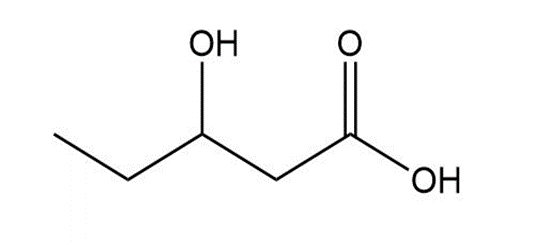
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.