Chapter 15: Q48. (page 570)
Question:Draw the products formed in each reaction and include the stereochemistry around any stereogenic centers.
a.
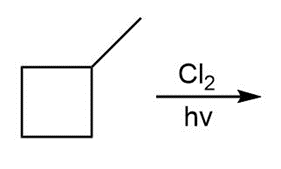
b.
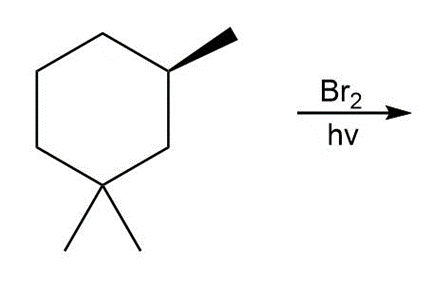
c.
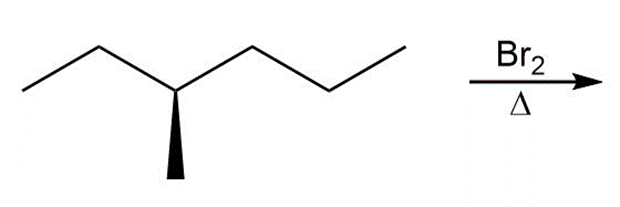
d.
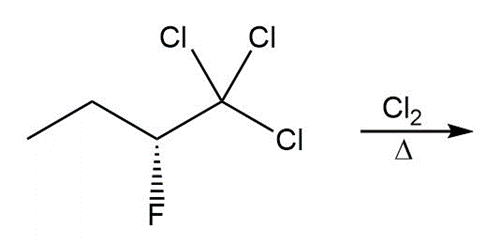
Short Answer
Answer
a.
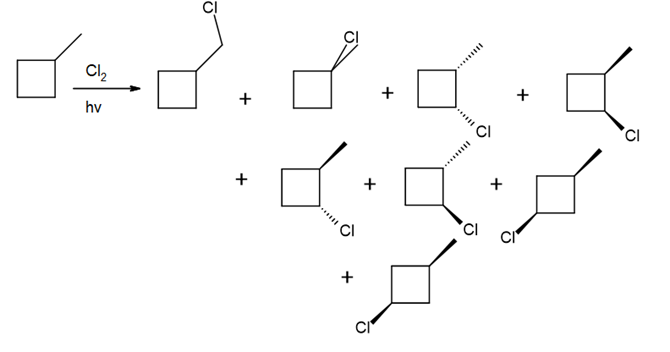
b.
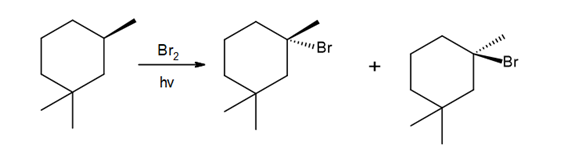
c.
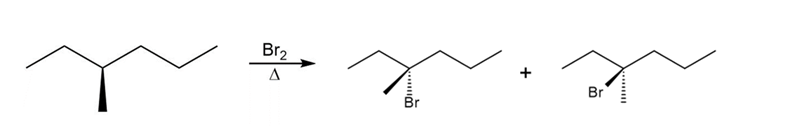
d.
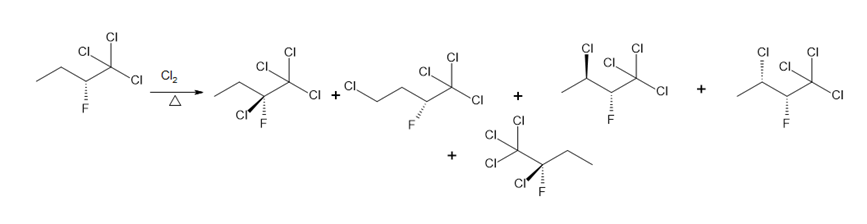
Learning Materials
Features
Discover
Chapter 15: Q48. (page 570)
Question:Draw the products formed in each reaction and include the stereochemistry around any stereogenic centers.
a.

b.

c.

d.

Answer
a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
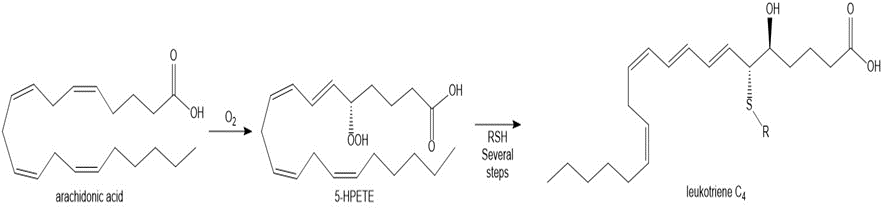
Question: As described in Section 9.16, the leukotrienes, important components in the asthmatic response, are synthesized from arachidonic acid via the hydroperoxide 5-HPETE. Write a stepwise mechanism for the conversion of arachidonic acid to 5-HPETE with O2 .
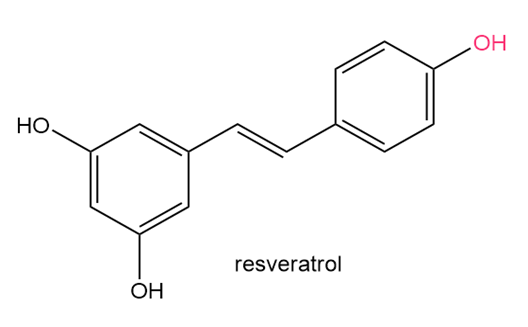
Question: Resveratrol is an antioxidant found in the skin of red grapes. Its anticancer, anti-inflammatory,and various cardiovascular effects are under active investigation. (a) Draw all resonancestructures for the radical that results from homolysis of the OH bond shown in red. (b) Explainwhy homolysis of this OH bond is preferred to homolysis of either OH bond in the otherbenzene ring.
Question: Draw the products formed when a chlorine atom reacts with each species.
a.
b.
c.
d. O2
Question: Draw the monochlorination products formed when each compound is heated with Cl2. Include the stereochemistry at any stereogenic center.
a.
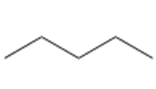
b.
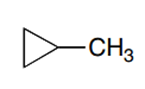
c.
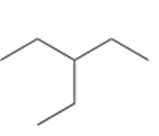
d.
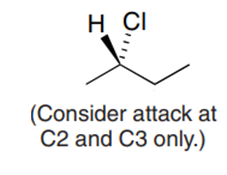
Question: What products are formed from monochlorination of (R)-2-bromobutane at C1 and C4? Assign R and S designations to each stereogenic center.
What do you think about this solution?
We value your feedback to improve our textbook solutions.