Chapter 15: Q44. (page 570)
Question: Draw all constitutional isomers formed when X is treated with NBS + hv.
Short Answer
Answer
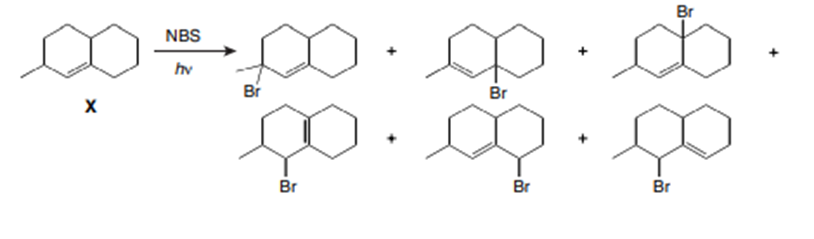
Isomers formed from compound X
Learning Materials
Features
Discover
Chapter 15: Q44. (page 570)
Question: Draw all constitutional isomers formed when X is treated with NBS + hv.

Answer

Isomers formed from compound X
All the tools & learning materials you need for study success - in one app.
Get started for free
Question:(a) Draw all stereoisomers of molecular formula C5H10Cl2 formed when (R)-2-chloropentane is heated with Cl2 .
(b) Assuming that products having different physical properties can beseparated into fractions by some physical method (such as fractional distillation), how many different fractions would be obtained?
(c) Which of these fractions would be optically active?
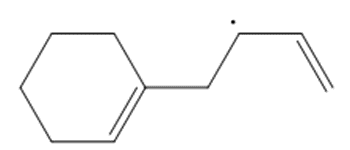
Question: Draw a second resonance structure for each radical. Then draw the hybrid.
a.
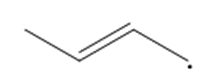
b.
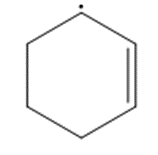
c.
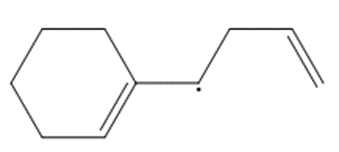
d.
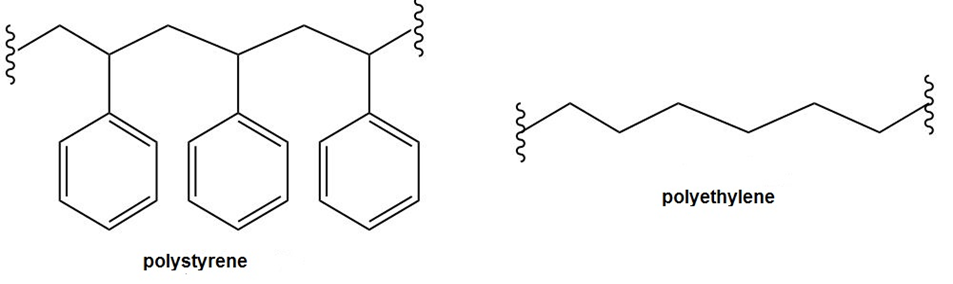
Question: Explain why polystyrene is much more readily oxidized by O2 in the air than polyethylene is. Which H’s in polystyrene are most easily abstracted and why?
Question:What reagents are needed to convert cyclopentene into
(a)bromocyclopentane;
(b) trans-1,2-dibromocyclopentane;
(c) 3-bromocyclopentene?
Question: Devise a synthesis of each compound using CH3CH3 as the only source of carbon atoms. You may use any other required organic or inorganic reagents.
a.
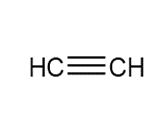
b.
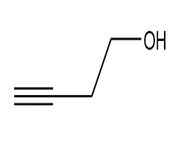
c.
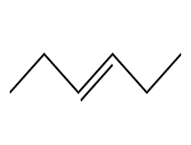
d.
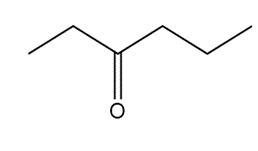
What do you think about this solution?
We value your feedback to improve our textbook solutions.