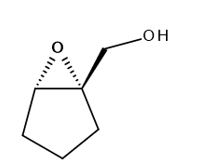
Chapter 12: PROBLEM 12.29 (page 487)
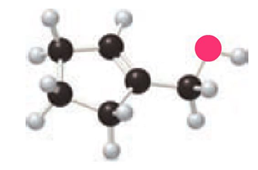
Question: Draw the products formed when A is treated with each reagent: (a) H2 Pd-C; (b) mCPBA; (c) PCC; (d) CrO3, H2SO4, H2O; (e) Sharpless reagent with (+)-DET.
Short Answer
Answer
a.
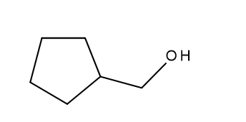
b.
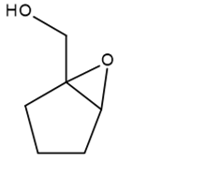
c.
d.
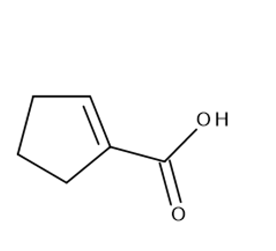
e.
Learning Materials
Features
Discover
Chapter 12: PROBLEM 12.29 (page 487)
Question: Draw the products formed when A is treated with each reagent: (a) H2 Pd-C; (b) mCPBA; (c) PCC; (d) CrO3, H2SO4, H2O; (e) Sharpless reagent with (+)-DET.

Answer
a.

b.

c.
d.

e.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Match each alkene to its heat of hydrogenation.Alkenes: 3-methylbut-1-ene, 2-methylbut-1-ene, 2-methylbut-2-ene (hydrogenation) kJ/mol: –119, –127, –112
Question: Which alkyne has the smaller Heat of hydrogenation ? Explain your choice.
Question: Draw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3+H2O. Explain how these reactions illustrate that syndihydroxylation is stereospecific.
Question: One compound that contributes to the “seashore smell”at beaches in Hawai’s is dictyopterene D’ with excess in the presence of a Pd catalyst forms butylcycloheptane. Ozonolysis with followed by forms ,, and . What are possible structures of dictyopterene D’?
Question: Devise a synthesis of each compound from the indicated starting material, organic compounds containing one or two carbons, and any other required reagents.
What do you think about this solution?
We value your feedback to improve our textbook solutions.