Chapter 12: PROBLEM 12.27 (page 484)
Question: Draw the products of each Sharpless epoxidation.
a.
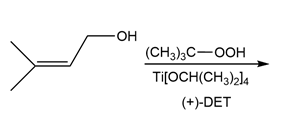
b.
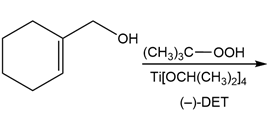
Short Answer
Answer
a.
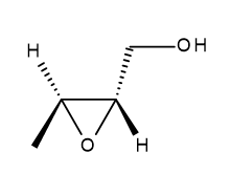
b.
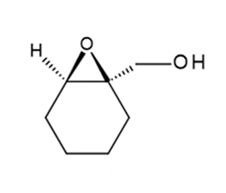
Learning Materials
Features
Discover
Chapter 12: PROBLEM 12.27 (page 484)
Question: Draw the products of each Sharpless epoxidation.
a.

b.

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Match each alkene to its heat of hydrogenation.Alkenes: 3-methylbut-1-ene, 2-methylbut-1-ene, 2-methylbut-2-ene (hydrogenation) kJ/mol: –119, –127, –112
Question: Devise a synthesis of each compound from the indicated starting material, organic compounds containing one or two carbons, and any other required reagents.
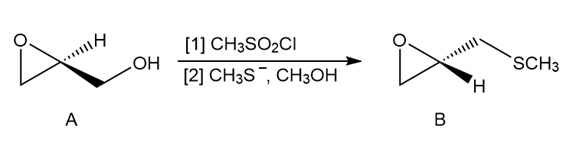
Question: a) What product is formed in Step [1] of the following reaction sequence?
(b) Draw a mechanism for Step [2] that accounts for the observed stereochemistry.
(c) What reaction conditions are necessary to form chiral A from prop-2-en-1-ol
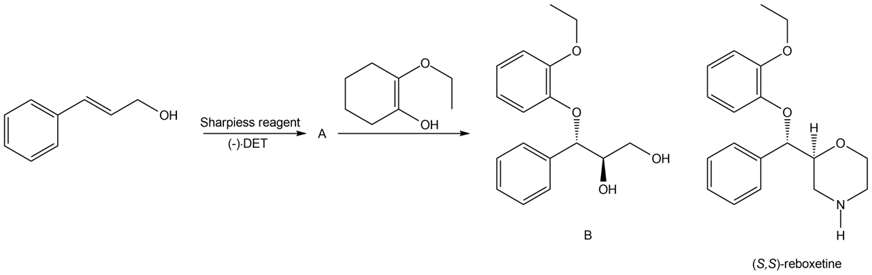
Question: Identify A in the following reaction sequence and draw a mechanism for the conversion of A to B. B has been converted to (S,S)-reboxetine, an antidepressant marketed outside the United States.
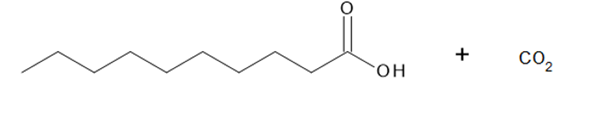
Question: What alkyne (or diyne) yields each set of oxidative cleavage products?
a.
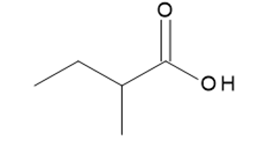
b.
c.
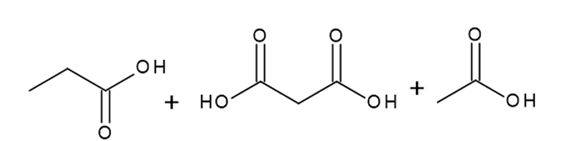
d.
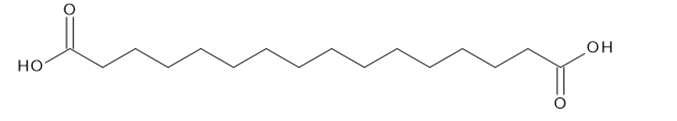
What do you think about this solution?
We value your feedback to improve our textbook solutions.