Chapter 14: Q6P (page 527)
Question: Label the protons in each highlighted \({\bf{C}}{{\bf{H}}_{\bf{2}}}\) group as enantiotopic, diastereotopic, or homotopic.
a. 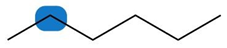
b. 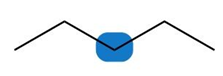
c. 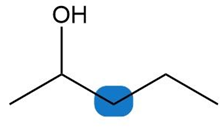
Short Answer
Enantiotopic
Neither
Diastereotopic
Learning Materials
Features
Discover
Chapter 14: Q6P (page 527)
Question: Label the protons in each highlighted \({\bf{C}}{{\bf{H}}_{\bf{2}}}\) group as enantiotopic, diastereotopic, or homotopic.
a. 
b. 
c. 
All the tools & learning materials you need for study success - in one app.
Get started for free
a.
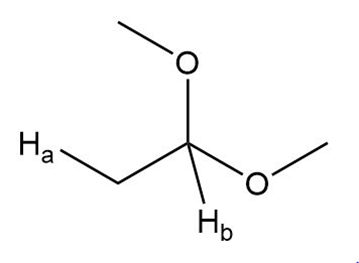
b.
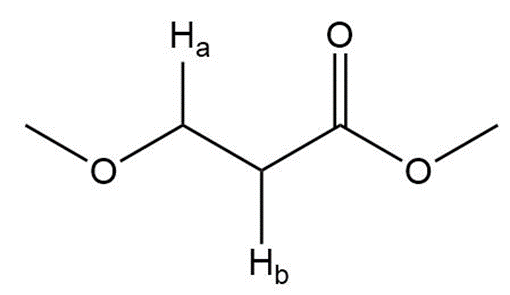
c.
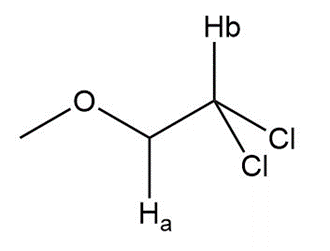
d.
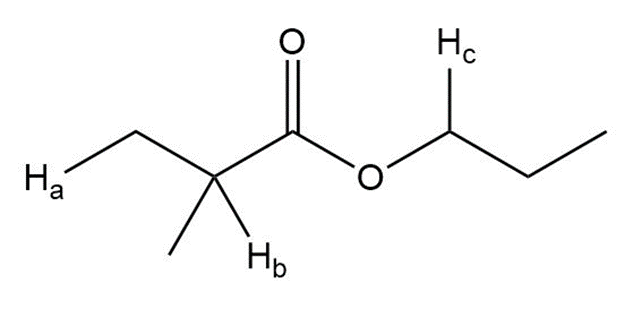
e.
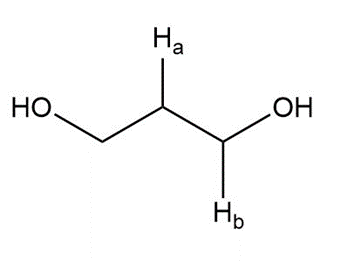
f.
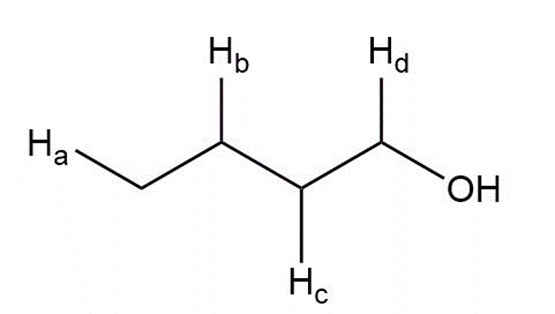
g.
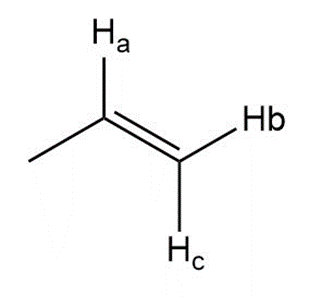
h.
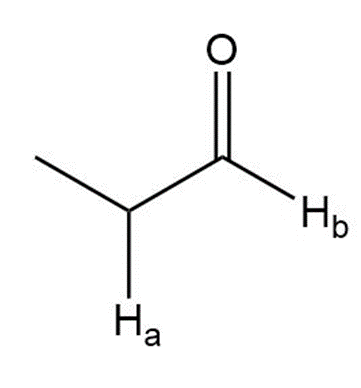
i.
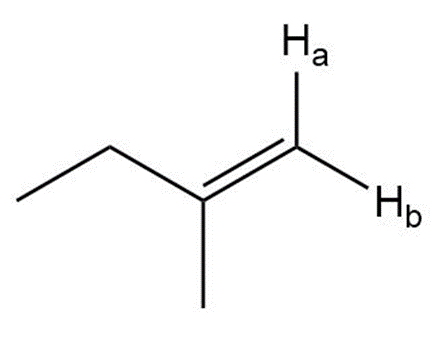
j.
The 1H NMR spectrum 1,2-dimethoxyethane (CH3OCH2CH2OCH3) recorded on a 300 MHz NMR spectrometer consists of signals at 1017 Hz and 1065 Hz downfield from TMS. (a) Calculate the chemical shift of each absorption. (b) At what frequency would each absorption occur if the spectrum were recorded on a 500 MHz NMR spectrometer?
The reaction of (CH3)3 CCHO with (C6H5)3 P=C(CH3)OCH3 , followed by treatment with aqueous acid, afford R . R has strong absorption in its IR spectrum at 1717 cm-1 and three singlets in its -NMR spectrum at 1.02 (9 H), 2.13 (3 H), and 2.33 (2 H) ppm. What is the structure of R? We will learn about this reaction in Chapter 21.
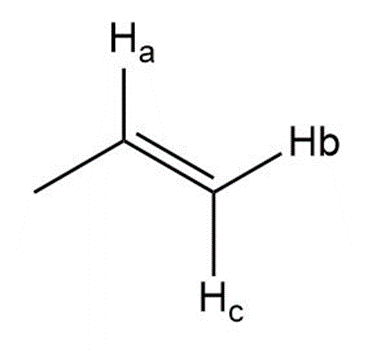
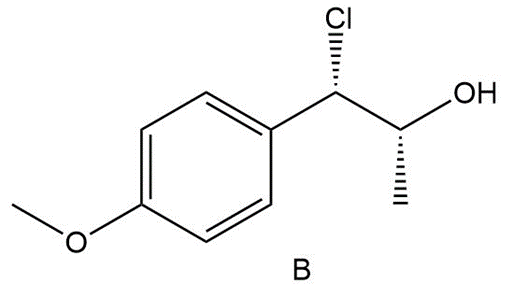
Question:Reaction of unknown A with HCl forms chlorohydrin B as the major product. A shows no absorptions in its IR spectrum at 1700 cm-1 or 3600-3200 cm-1 , and gives the following 1H NMR data: 1.4 (doublet, 3 H), 3.0 (quartet of doublets, 1 H), 3.5 (doublet, 1 H), 3.8 (singlet, 3 H), 6.9 (doublet, 2 H), and 7.2 (doublet, 2 H) ppm.
(a) Propose a structure for A, including stereochemistry.
(b) Explain why B is the major product in this reaction.
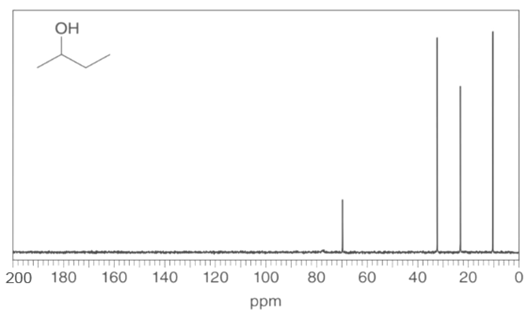
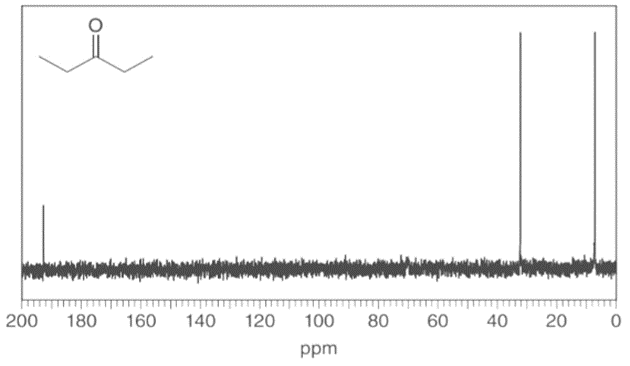
Question: Identify the carbon atoms that give rise to each NMR signal.
a.
b
What do you think about this solution?
We value your feedback to improve our textbook solutions.