Chapter 14: Q56P (page 527)
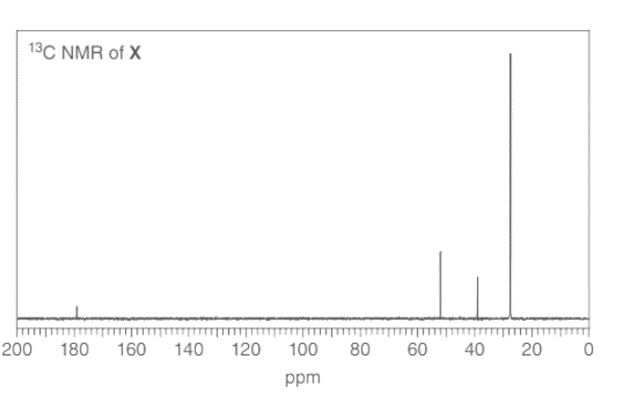
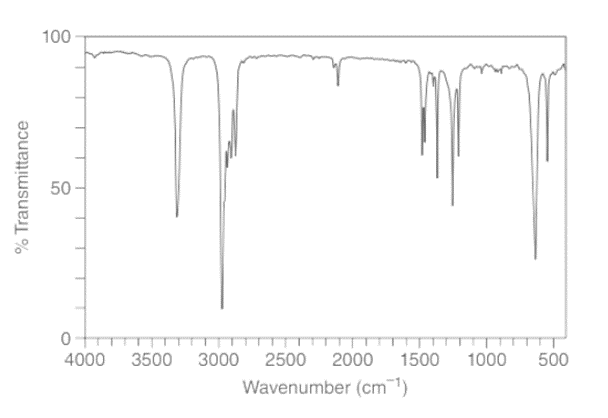
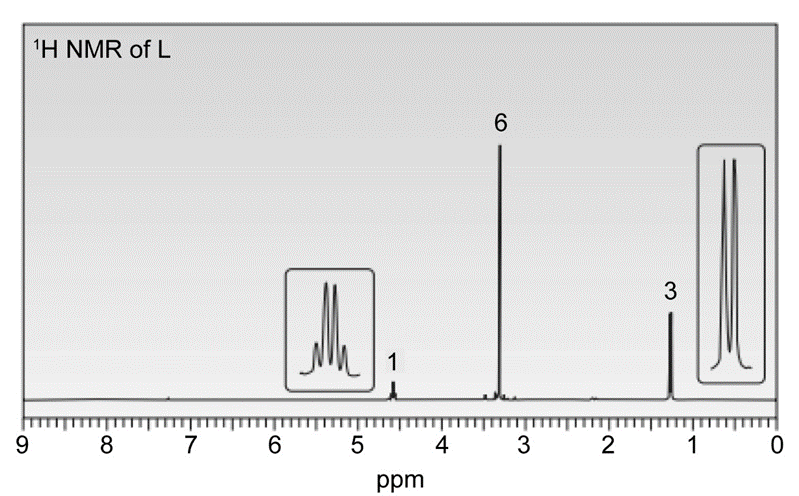
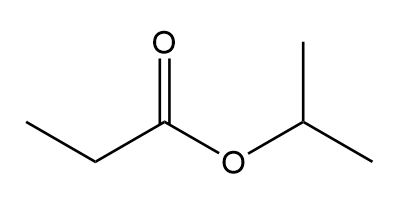
Treatment of 2-methylpropanenitrile \(\left( {{{\left( {{\bf{C}}{{\bf{H}}_{\bf{3}}}} \right)}_{\bf{2}}}{\bf{CHCN}}} \right)\) with\({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{MgBr}}\), followed by aqueous acid, affords compound V, which has molecular formula \({{\bf{C}}_{\bf{7}}}{{\bf{H}}_{{\bf{14}}}}{\bf{O}}\). V has a strong absorption in its IR spectrum at 1713 \({\rm{c}}{{\rm{m}}^{{\rm{ - 1}}}}\), and gives the following 1 H NMR data: 0.91 (triplet, 3 H), 1.09 (doublet, 6 H), 1.6 (multiplet, 2 H), 2.43 (triplet, 2 H), and 2.60 (septet, 1 H) ppm. What is the structure of V? We will learn about this reaction in Chapter 22.
Short Answer