Chapter 14: Q.21558-14-7P. (page 535)
How many NMR signals would you expect for each compound?
a.
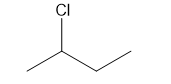
b.
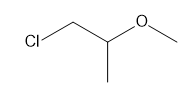
c.
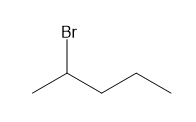
Short Answer
a. 4
b. 5
c. 5
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-7P. (page 535)
How many NMR signals would you expect for each compound?
a.

b.

c.

a. 4
b. 5
c. 5
All the tools & learning materials you need for study success - in one app.
Get started for free
For each compound, first label each different type of proton and then rank the protons in order of increasing chemical shift.
a.
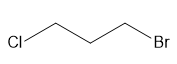
b.
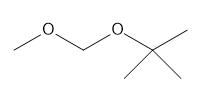
c.
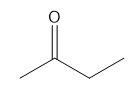
Question: As we will learn in Chapter 20, reaction of (CH3)2CO with followed by affords compound D, which has a molecular ion in its mass spectrum at 84 and prominent absorptions in its IR spectrum at 3600–3200, 3303, 2938, and 2120cm-1 . D shows the following 1 H NMR spectral data: 1.53 (singlet, 6 H), 2.37 (singlet, 1 H), and 2.43 (singlet, 1 H) ppm. What is the structure of D?
Question: Explain why the 13C NMR spectrum of 3-methylbutan-2-ol shows five signals.
Question: Cyclohex-2-enone has two protons on its carbon-carbon double bond (labeled Ha and Hb ) and two protons on the carbon adjacent to the double bond (labeled Hc ). (a) If Jab = 11 Hz and Jbc = 4 Hz, sketch the splitting pattern observed for each proton on the hybridized carbons. (b) Despite the fact that Ha is located adjacent to an electron-withdrawing C=O, its absorption occurs up-field from the signal due to Hb(6.0 vs. 7.0 ppm). Offer an explanation.
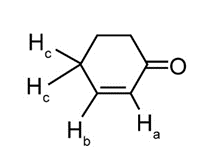
What do you think about this solution?
We value your feedback to improve our textbook solutions.