Chapter 31: Q.26. (page 1231)
Question: Locate the isoprene units in each compound.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 31: Q.26. (page 1231)
Question: Locate the isoprene units in each compound.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products formed when triacylglycerol A is treated with each reagent. Rank compounds A, B and C in order of increasing melting point.
Question: An isoprene unit can be thought of as having a head and a tail. The “head” of the isoprene unit is located at the end of the chain nearest the branch point, and the “tail” is located at the end of the carbon chain farthest from the branch point. Most isoprene units are connected together in a “head-to-tail” fashion, as illustrated. For both lycopene (Problem 31.26), and squalene (Figure 31.9), decide which isoprene units are connected in a head-to-tail fashion and which are not.
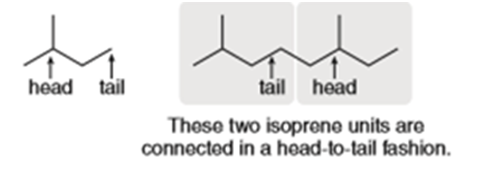
Locate the isoprene units in biformene, a component of amber, and classify biformene as a monoterpene, sesquiterpene etc.
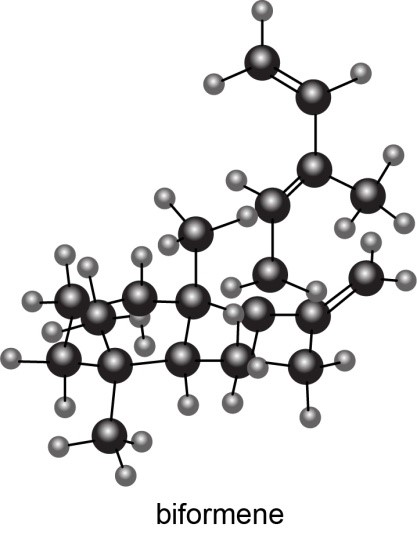
Question: Draw a stepwise mechanism for the conversion of neryl diphosphate to , is a component of pine oil and rosemary oil.
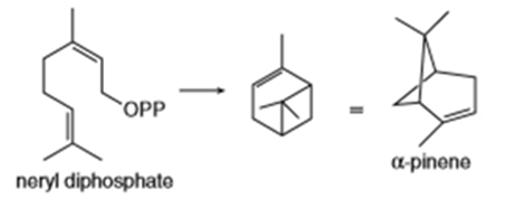
Locate the isoprene units in each compound.
a.
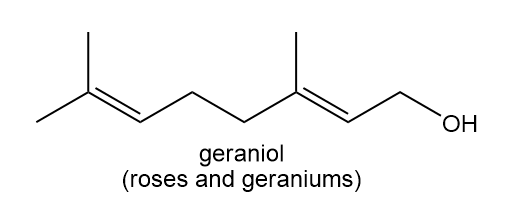
b.
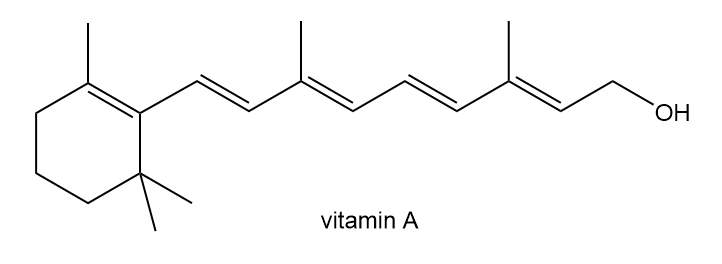
c.
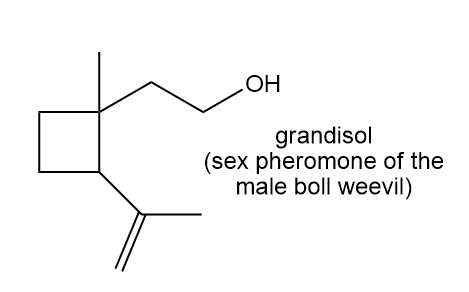
d.
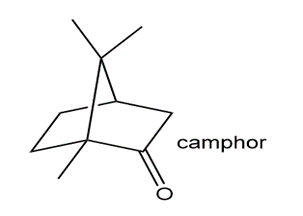
What do you think about this solution?
We value your feedback to improve our textbook solutions.