Chapter 20: Q57 (page 812)
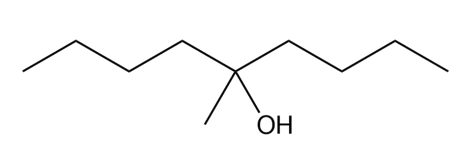
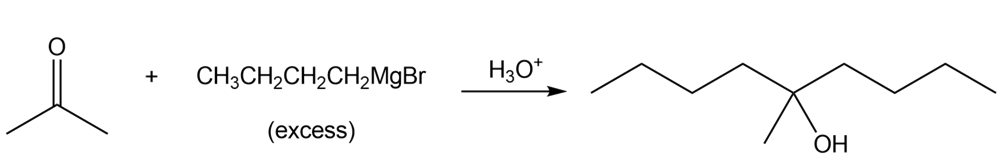
What ester and Grignard reagent are needed to synthesize each alcohol?
a.

b.
Short Answer
a.

b.
Learning Materials
Features
Discover
Chapter 20: Q57 (page 812)
What ester and Grignard reagent are needed to synthesize each alcohol?
a.

b.

a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
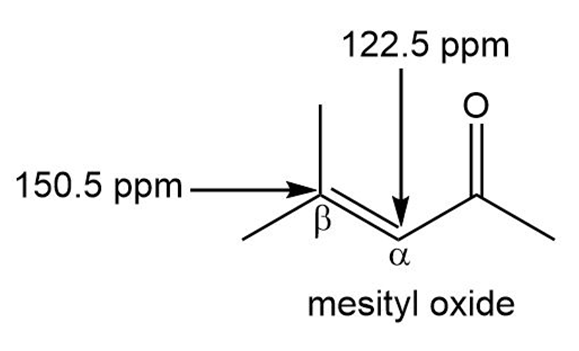
Explain why the carbon of an ,-unsaturated carbonyl compound absorbs farther downfield in the NMR spectrum than the a carbon, even though the carbon is closer to the electron-withdrawing carbonyl group. For example, the carbon of mesityl oxide absorbs at 150.5 ppm, while the carbon absorbs at 122.5 ppm.
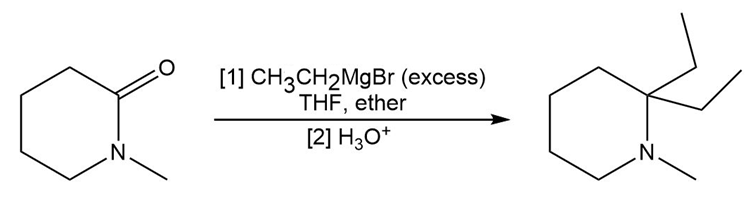
Draw a stepwise mechanism for the following reaction of a Grignard reagent with a cyclic amide.
Convert propan-2-ol into each compound. You may use any other organic or inorganic compounds
a.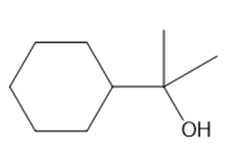
b.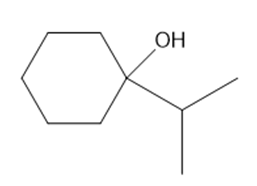
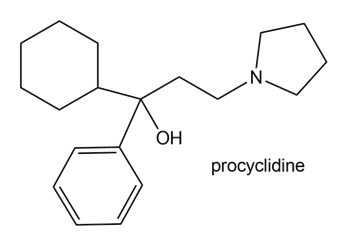
Procyclidine is a drug that has been used to treat the uncontrolled body movements associated with Parkinson’s disease. Draw three different methods to prepare procyclidine using a Grignard reagent
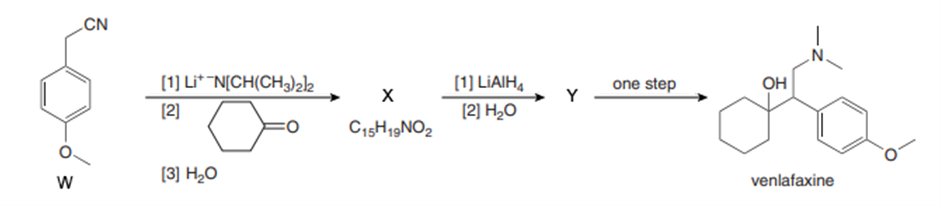
Identify X and Y, two of the intermediates in a synthesis of the antidepressant venlafaxine (trade name Effexor), in the following reaction scheme. Write a mechanism for the formation of X from W.
What do you think about this solution?
We value your feedback to improve our textbook solutions.