Chapter 20: Q41 (page 809)
Draw the product formed when the -unsaturated ketone A is treated with each reagent.
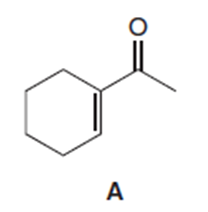
Short Answer
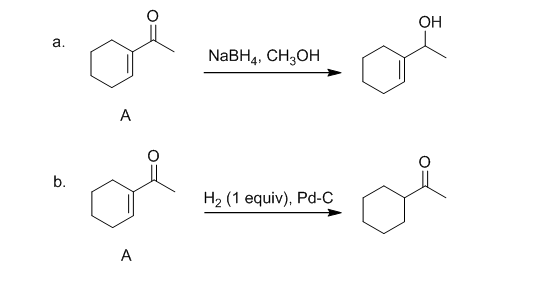
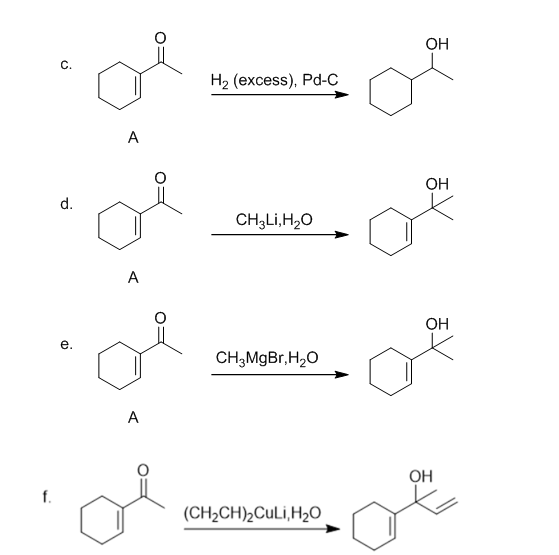
The products formed when structure A is treated with the mentioned reagents are shown below:-
Learning Materials
Features
Discover
Chapter 20: Q41 (page 809)
Draw the product formed when the -unsaturated ketone A is treated with each reagent.

The products formed when structure A is treated with the mentioned reagents are shown below:-


All the tools & learning materials you need for study success - in one app.
Get started for free
Treatment of isobutene with forms a carbanion that reacts with to form H after water is added to the reaction mixture. H has a molecular ion in its mass spectrum at , and shows fragments at 71 and 68. H exhibits absorptions in its IR spectrum at 3600–3200 and 1651 , and has the NMR spectrum given below. What is the structure of H?
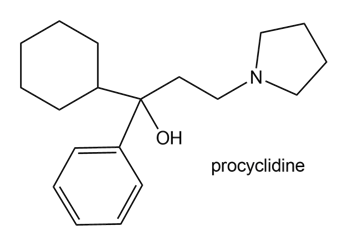
Procyclidine is a drug that has been used to treat the uncontrolled body movements associated with Parkinson’s disease. Draw three different methods to prepare procyclidine using a Grignard reagent
Design a synthesis of each compound from alcohols having four carbons or fewer as the only organic starting materials. You may use any other inorganic reagents you choose.
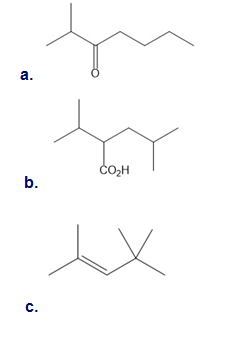
Devise a synthesis of the given alcohol from benzene, organic alcohols having four or fewer carbons, and any needed inorganic reagents.
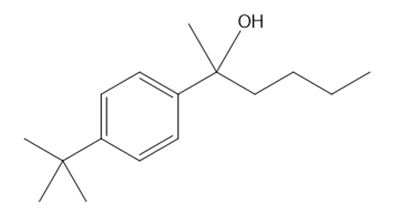
Draw all stereoisomers formed in each reaction.

What do you think about this solution?
We value your feedback to improve our textbook solutions.