Chapter 22: Q21. (page 896)
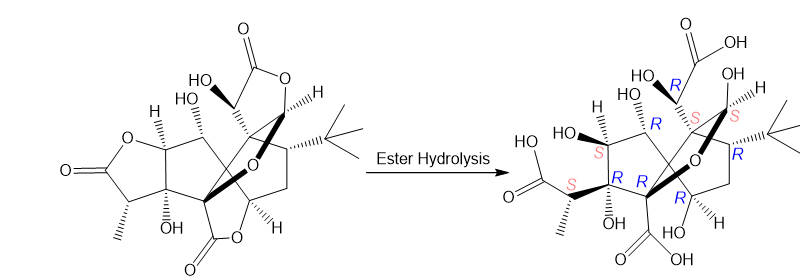
Question: What product is formed when the esters in ginkgolide B, the chapter-opening molecule, are hydrolyzed in aqueous acid? Indicate the stereochemistry of all stereogenic centers.

Short Answer
Answer
Learning Materials
Features
Discover
Chapter 22: Q21. (page 896)
Question: What product is formed when the esters in ginkgolide B, the chapter-opening molecule, are hydrolyzed in aqueous acid? Indicate the stereochemistry of all stereogenic centers.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question:What reagents are needed to convert phenylacetonitrile () to each compound:
stereochemistry of the products of reduction depends on the reagent used,as you learned in Sections 20.5 and 20.6. With this in mind, how would you convert3,3-dimethylbutan-2-one [CH3COC(CH3)3] to: (a) racemic 3,3-dimethylbutan-2-ol[CH3CH(OH)C (CH3)3] ;(b) only (R)-3,3-dimethylbutan-2-ol; (c) only (S)-3,3-dimethylbutan-2-ol?
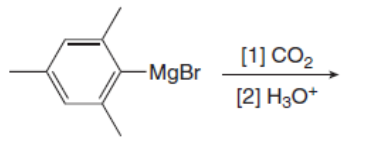
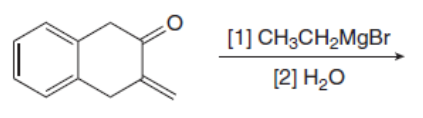
Draw the products of the following reactions with organometallic reagents.
a.
b.
c.
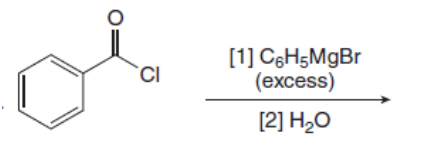
d.
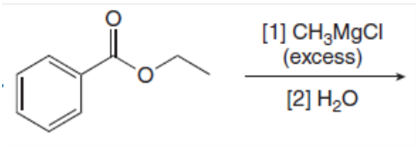
e.
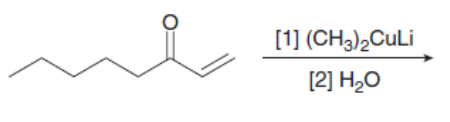
f.
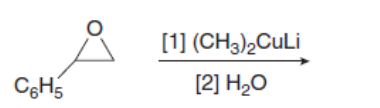
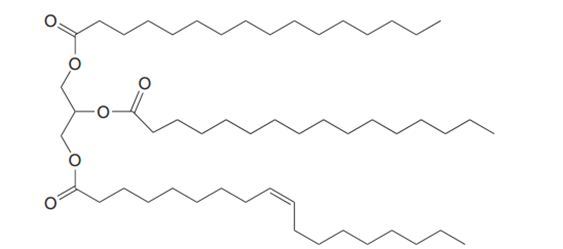
Question: What is the composition of the soap prepared by hydrolysis of the following triacylglycerol?
What Grignard reagent and carbonyl compound can be used to prepare the antidepressant venlafaxine (trade name Effexor)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.