Chapter 22: 19P (page 868)
Which of the following species represent organometallic compounds:
(a)BrMgCCCH2CH3 ; (b) NaOCH2CH3 ; (c)KOC(CH3)3 ; (d) PhLi?
Short Answer
Answer
(a)BrMgC CCH2CH3 and (d)PhLi represent an organometallic compound.
Learning Materials
Features
Discover
Chapter 22: 19P (page 868)
Which of the following species represent organometallic compounds:
(a)BrMgCCCH2CH3 ; (b) NaOCH2CH3 ; (c)KOC(CH3)3 ; (d) PhLi?
Answer
(a)BrMgC CCH2CH3 and (d)PhLi represent an organometallic compound.
All the tools & learning materials you need for study success - in one app.
Get started for free
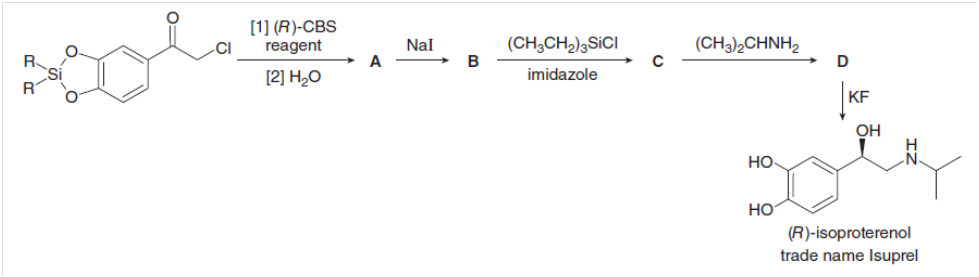
Fill in the lettered products (A–D) in the synthesis of (R)-isoproterenol, a drug that increases heart rate and dilates lung passages.
Question: Explain why trichloroacetic anhydride is more reactive than acetic anhydride in nucleophilic acyl substitution reactions.
Review the oxidation reactions using Cr+6 reagents in Section 12.12. Then, draw the product formed when compound B is treated with each reagent.
a. NaBH4, CH3OH
b. [1] LiAlH4 ; [2]H2O
c. PCC
d.Ag2O, NH4OH
e. CrO3, H2SO4, H2O
Draw the product formed when pentanal (CH3CH2CH2CH2CHO ) is treated with each reagent. With some reagents, no reaction occurs.
a. NaBH4 , CH3OH
b. {1} LiAlH4 ; {2} H2O
c. H2,Pd-C
d. PCC
e. Na2Cr2O7, H2SO4, H2O
f. Ag2O, NH4OH
g. {1}CH3MgBr ; {2} H2O
h. {1} C6H5Li ; {2} H2O
i. {1} (CH3)2CuLi ; {2} H2O
l. The product in (a) , then TBDMS-Cl , imidazole
Question: Give an IUPAC or common name for each compound.
a.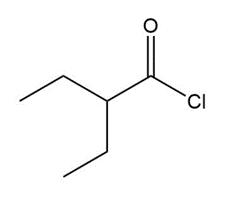
b. 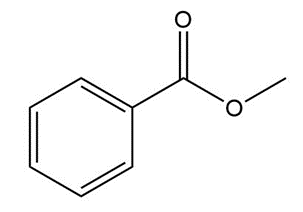
c. 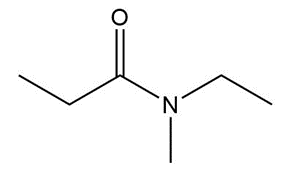
d. 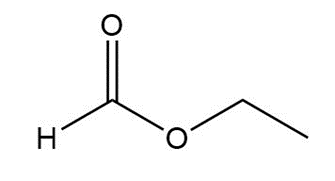
e. 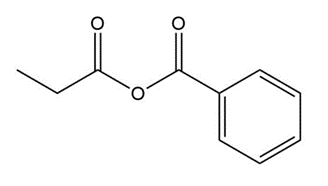
f. 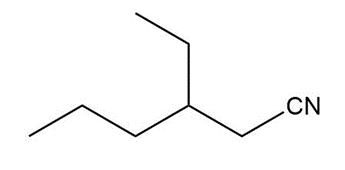
What do you think about this solution?
We value your feedback to improve our textbook solutions.