Chapter 22: 10P (page 868)
Draw a stepwise mechanism for the following reaction.
Short Answer
Answer
The mechanism shown for the reduction of acid chloride by LiAlH4 to alcohol is shown below:
Learning Materials
Features
Discover
Chapter 22: 10P (page 868)
Draw a stepwise mechanism for the following reaction.
Answer
The mechanism shown for the reduction of acid chloride by LiAlH4 to alcohol is shown below:
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Fenofibric is a cholesterol-lowering medication that is converted to fenofibric acid, the active drug, by hydrolysis during metabolism. What is the structure of fenofibric acid?
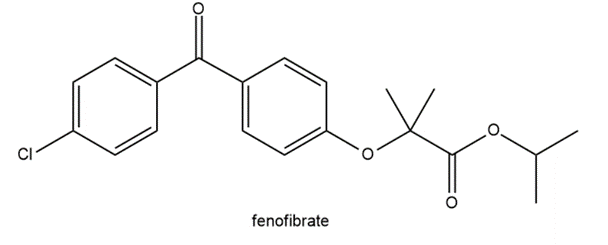
Draw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs.
Question:Outline two different ways that butan-2-one can be prepared from a nitrile and a Grignard reagent.
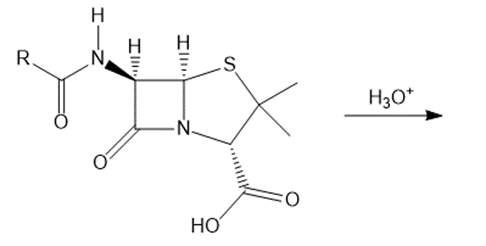
Question: Some penicillins cannot be administered orally because their β-lactam is rapidly hydrolyzed by the acidic environment of the stomach. What product is formed in the following hydrolysis reaction?
stereochemistry of the products of reduction depends on the reagent used,as you learned in Sections 20.5 and 20.6. With this in mind, how would you convert3,3-dimethylbutan-2-one [CH3COC(CH3)3] to: (a) racemic 3,3-dimethylbutan-2-ol[CH3CH(OH)C (CH3)3] ;(b) only (R)-3,3-dimethylbutan-2-ol; (c) only (S)-3,3-dimethylbutan-2-ol?
What do you think about this solution?
We value your feedback to improve our textbook solutions.