Chapter 19: Q69 (page 763)
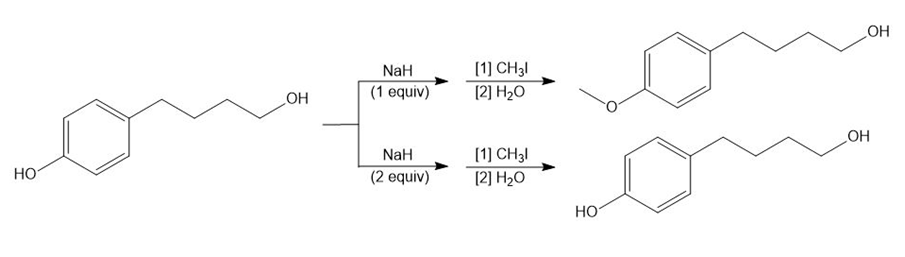
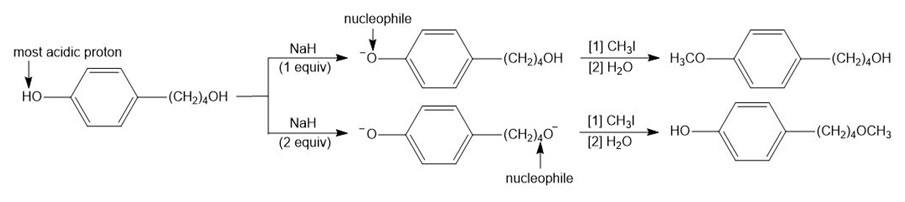
Explain why using one or two equivalents of NaH results in different products in the following reactions.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q69 (page 763)
Explain why using one or two equivalents of NaH results in different products in the following reactions.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Draw the structure corresponding to each name.
For each amino acid , draw its neutral, positively charged, and negatively charged forms. Which form predominates at pH = 1, 6, and 11? What is the structure of each amino acid at its isoelectric point?
a. methionine
b. serine
Question: Explain why theof the COOH group of glycine is much lower than the of the COOH of acetic acid (2.35 compared to 4.8).
Question: Give the structure corresponding to each IUPAC name.
a. 2-bromobutanoic acid
b. 2,3-dimethylpentanoic acid
c. 3,3,4-trimethylheptanoic acid
d. 2-sec-butyl-4,4-diethylnonanoic acid
e. 3,4-diethylcyclohexanecarboxylic acid
f. 1-isopropylcyclobutanecarboxylic acid
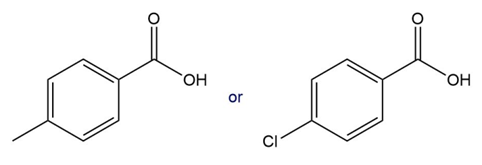
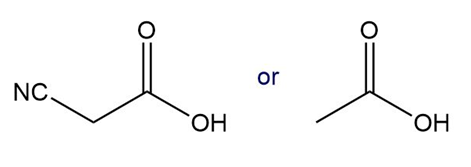
Which compound in each pair has the lower ? Which compound in each pair has the stronger conjugate base?
a.
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.