Chapter 19: Q14. (page 744)
Question: Given the values in Appendix A, which of the following bases are strong enough to deprotonate : (a) F ; (b) ; (c) ?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 19: Q14. (page 744)
Question: Given the values in Appendix A, which of the following bases are strong enough to deprotonate : (a) F ; (b) ; (c) ?
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Rank the following compounds in order of increasing boiling point. Which compound is the most water soluble? Which compound is the least water soluble?
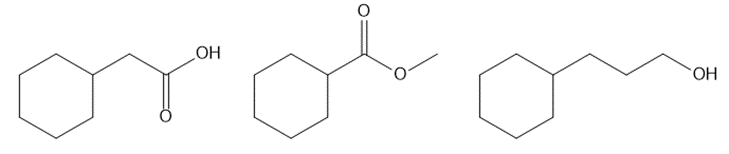
Question: Draw the organic products formed in each reaction.
a. 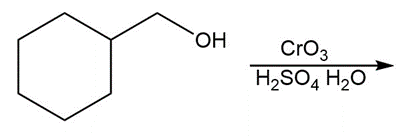
b.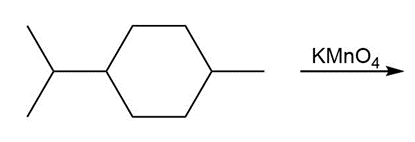
c. 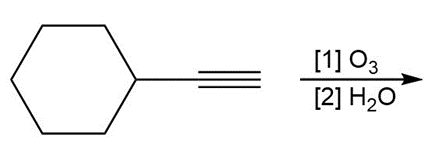
d.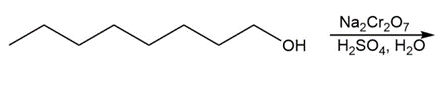
Question: Draw the structure corresponding to each name.
For each amino acid , draw its neutral, positively charged, and negatively charged forms. Which form predominates at pH = 1, 6, and 11? What is the structure of each amino acid at its isoelectric point?
a. methionine
b. serine
Question: The values for the carboxy and ammonium protons of phenylalanine are 2.58 and 9.24, respectively. What is the isoelectric point of phenylalanine? Draw the structure of phenylalanine at its isoelectric point.
What do you think about this solution?
We value your feedback to improve our textbook solutions.