Chapter 24: Q.52. (page 962)
Question: Draw a stepwise mechanism for the following variation of the aldol reaction, often called a nitro aldol reaction.
Short Answer
Answer
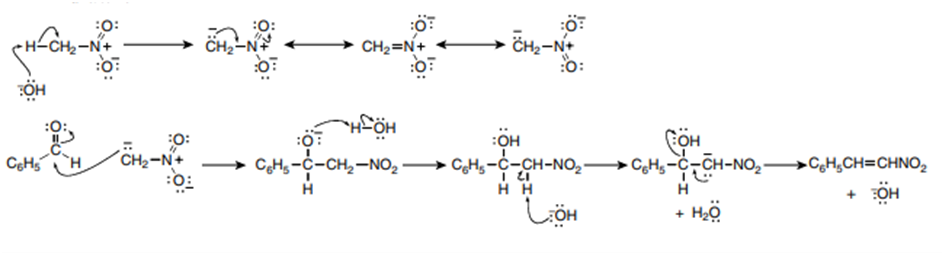
Mechanism
Learning Materials
Features
Discover
Chapter 24: Q.52. (page 962)
Question: Draw a stepwise mechanism for the following variation of the aldol reaction, often called a nitro aldol reaction.
Answer

Mechanism
All the tools & learning materials you need for study success - in one app.
Get started for free
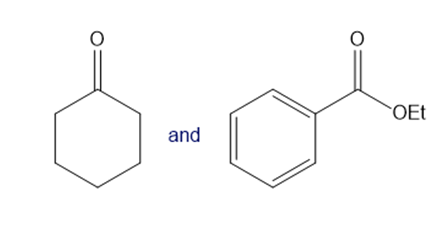
Question: What crossed Claisen product is formed from each pair of compounds?
a.
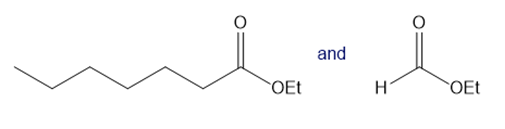
b.
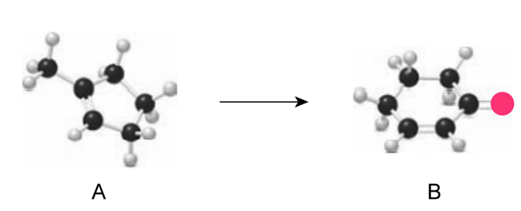
Question: Following the two-step reaction sequence depicted in Figure 24.5, write out the steps needed to convert A to B.
Question:Draw the product formed from a Claisen reaction with the given starting materials using –OEt, EtOH.
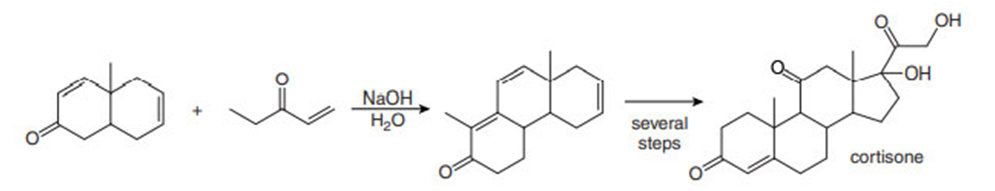
Question: Draw a stepwise mechanism for the following Robinson annulation. This reaction was a key step in a synthesis of the steroid cortisone by R. B. Woodward and co-workers at Harvard University in 1951.
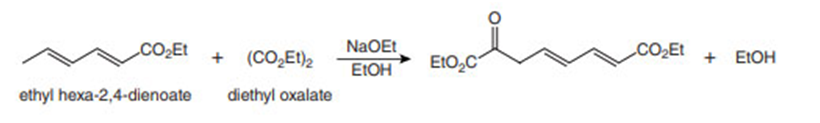
Question: (a) Draw a stepwise mechanism for the reaction of ethyl hexa-2,4-dienoate with diethyl oxalate in the presence of base.
(b) How does your mechanism explain why a new carbon-carbon bond forms on C6?
(c) Why is this reaction an example of a crossed Claisen reaction?
What do you think about this solution?
We value your feedback to improve our textbook solutions.