Chapter 24: Q.23. (page 962)
Question: What product is formed when each pair of compounds is treated with NaOEt in ethanol?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 24: Q.23. (page 962)
Question: What product is formed when each pair of compounds is treated with NaOEt in ethanol?
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What cyclic product is formed when each 1,5-dicarbonyl compound is treated with aqueous ?
Question: Acid-catalyzed dehydration of β-hydroxy carbonyl compounds occurs by the mechanism discussed in Section 9.8. With this in mind, draw a stepwise mechanism for the following reaction.
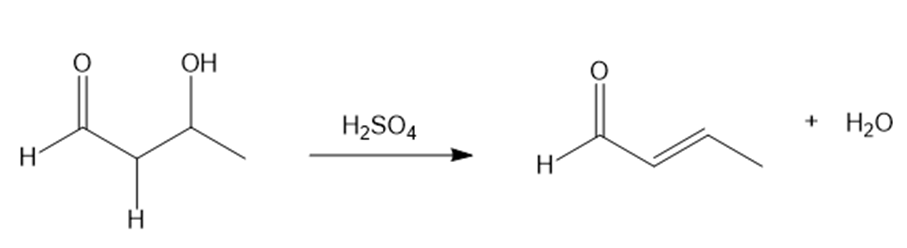
Question: What aldol product is formed when two molecules of butanal react together in the presence of base? What reagents are needed to convert this product to each of the following compounds?
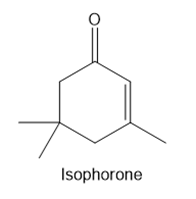
Question: Isophorone is formed from three molecules of acetone in the presence of base. Draw a mechanism for this process.
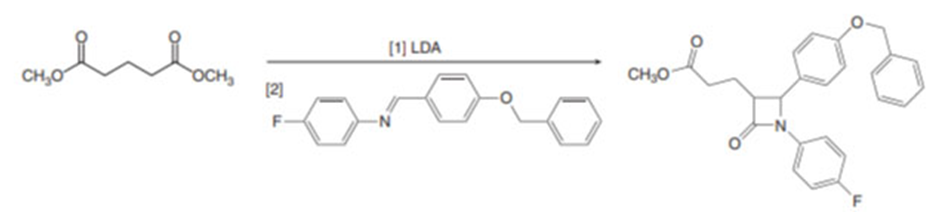
Question: Draw a stepwise mechanism for the following reaction, which was used in the synthesis of ezetimibe (Section 20.6), a drug used to treat patients with high cholesterol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.