Chapter 24: Q.13. (page 962)
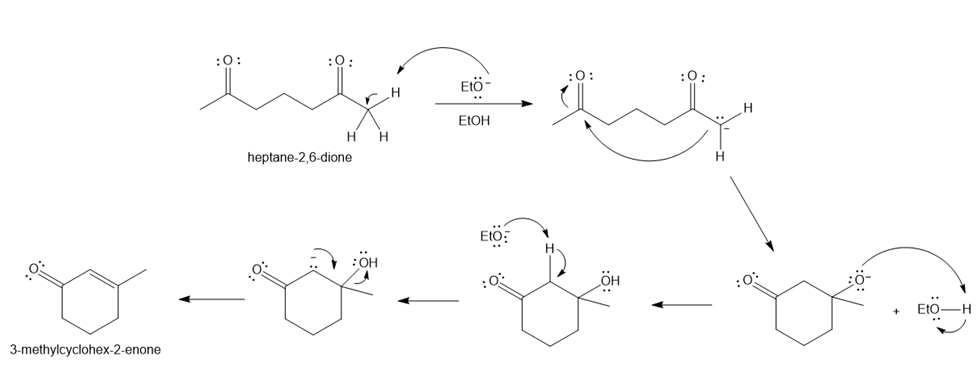
Question: Draw a stepwise mechanism for the conversion of heptane-2,6-dione to 3-methylcyclohex-2-enone with NaOEt, EtOH.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 24: Q.13. (page 962)
Question: Draw a stepwise mechanism for the conversion of heptane-2,6-dione to 3-methylcyclohex-2-enone with NaOEt, EtOH.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question:What starting materials are needed to prepare each compound using a Michael reaction?
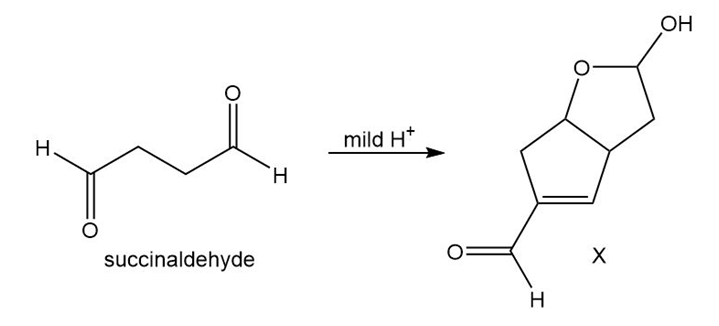
Question:One step in a recent short synthesis of a prostaglandin (Section 19.6) involves the conversion of succinaldehyde to the bicyclic hemiacetal X. Draw a stepwise mechanism for this process. (Hint: The mechanism begins with an intermolecular aldol reaction.)
Question: What starting materials are needed to synthesize each compound using an aldol or similar reaction?
Question: Answer the following questions about 2-acetylcyclopentanone.
a. What starting materials are needed to form 2-acetylcyclopentanone by a Claisen reaction that forms bond (a)?
b. What starting materials are needed to form 2-acetylcyclopentanone by a Claisen reaction that forms bond (b)?
c. What product is formed when 2-acetylcyclopentanone is treated with , followed by ?
d. Draw the Robinson annulation product(s) formed by reaction of 2-acetylcyclopentanone with methyl vinyl ketone .
e. Draw the structure of the most stable enol tautomer(s).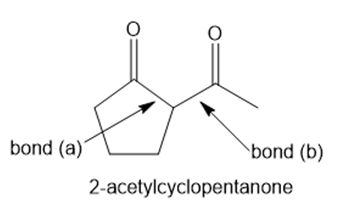
Question: Devise a synthesis of 2-methylcyclopentanone from cyclohexene. You may also use any required reagents
What do you think about this solution?
We value your feedback to improve our textbook solutions.