Chapter 28: Q3. (page 1106)
Question: Convert the ball-and-stick model to a Fischer projection.
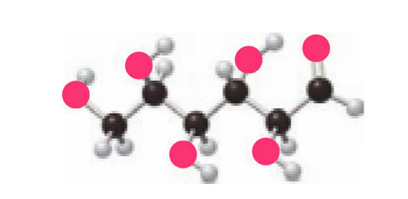
Short Answer
Answer
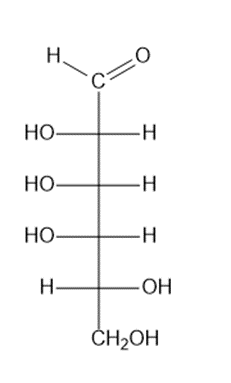
Fischer projection of given compound
Learning Materials
Features
Discover
Chapter 28: Q3. (page 1106)
Question: Convert the ball-and-stick model to a Fischer projection.

Answer

Fischer projection of given compound
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider the following six compounds (A-F).
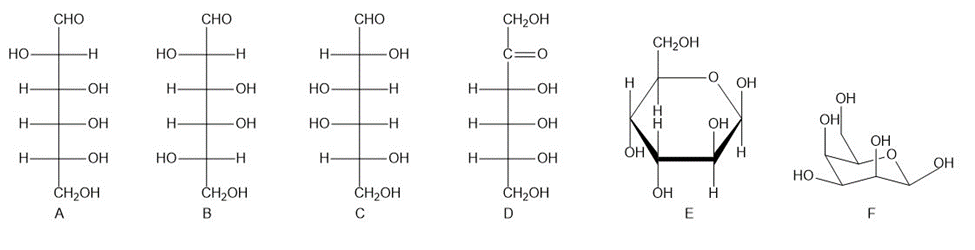
How are the two compounds in each pair related? Choose from enantiomers, epimers, diastereomers but not epimers, constitutional isomers, and identical compounds.
A and B
A and C
B and C
A and D
E and F
Question: Convert each aldohexose to the indicated anomer using a Haworth projection
Question: Label each stereogenic center as RorS.
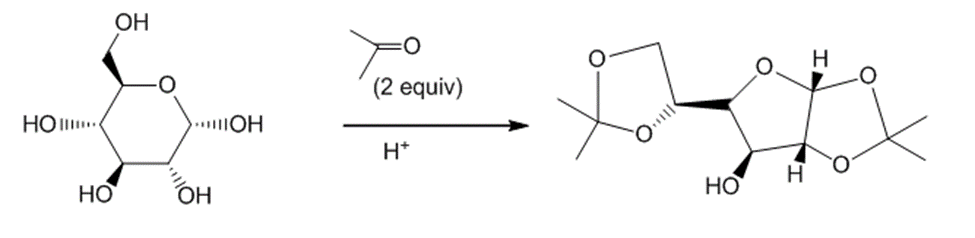
Draw a stepwise mechanism for the following reaction.
The most stable conformation of the pyranose ring of most D-aldohexoses places the largest group, , in the equatorial position. An exception to this is the aldohexose D-idose. Draw the two possible chair conformations of either the or anomers of D-idose. Explain why the more stable conformation has the group in the axial position.
What do you think about this solution?
We value your feedback to improve our textbook solutions.