Chapter 29: Q30P (page 1191)
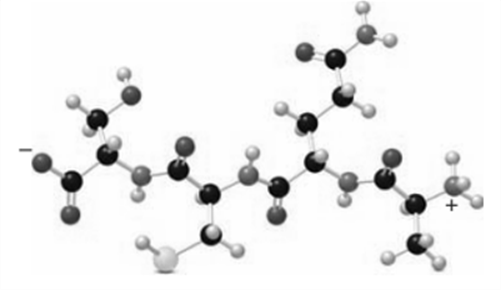
With reference to the following peptide:
(a) Identify the N-terminal and C-terminal amino acids.
(b) Name the peptide using one-letter abbreviations.
(c) Label all the amide bonds in the peptide backbone.
Short Answer
(a) The N-terminal amino acid is Alanine. In the given peptide, Alanine has a free anime group.
The N-terminal amino acid is Serine in the given peptide. It is the amino acid in which free carboxylic acid is present.
(b) The name of the peptide is Ser-Cys-Glu-Ala.
(c)
 Tetrapeptide
Tetrapeptide
 Tetrapeptide
Tetrapeptide Name of the peptide
Name of the peptide Tetrapeptide
Tetrapeptide
