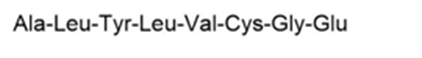Chapter 29: Q20. (page 1171)
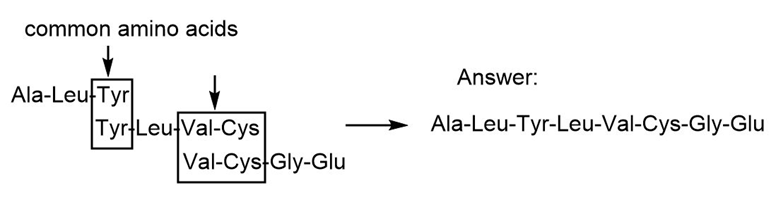
Give the amino acid sequence of an octapeptide that contains the amino acids Tyr, Ala, Leu (2 equiv), Cys, Gly, Glu and Val, and forms the following fragments when partially hydrolyzed with HCl: Val-Cys-Gly-Glu, Ala-Leu-Tyr, and Tyr-Leu-Val-Cys.
Short Answer
The amino acid sequence of an octapeptide is,