Chapter 25: PROBLEM 25.26 (page 1026)
Question: Draw the product formed by treating each compound with excess CH3I , followed by Ag2O , and then heat.
a.
b.
c.
Short Answer
Answer
a.CH3CH2CHCH2
b.CH3CHCH2
c.
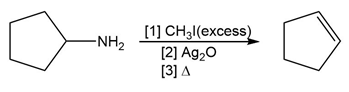
Learning Materials
Features
Discover
Chapter 25: PROBLEM 25.26 (page 1026)
Question: Draw the product formed by treating each compound with excess CH3I , followed by Ag2O , and then heat.
a.

b.

c.

Answer
a.CH3CH2CHCH2
b.CH3CHCH2
c.
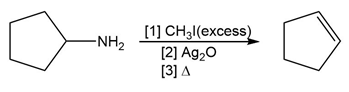
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: (a) What two components are needed to prepare para red by azo coupling? (b) What two components are needed to prepare alizarine yellow R?
Tertiary ( ) aromatic amines react with and HCl to afford products of electrophilic aromatic substitution. Draw a stepwise mechanism for this nitrosation reaction and explain why it occurs only on benzene rings with strong ortho, para activating groups.
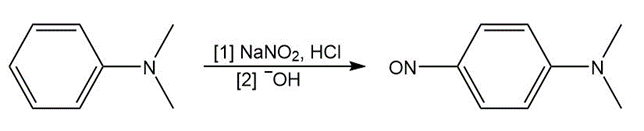
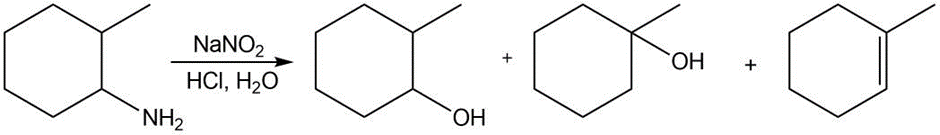
Alkyl diazonium salts decompose to form carbocations, which go on to form products of substitution, elimination, and (sometimes) rearrangement. Keeping this in mind, draw a stepwise mechanism that forms all of the following products.
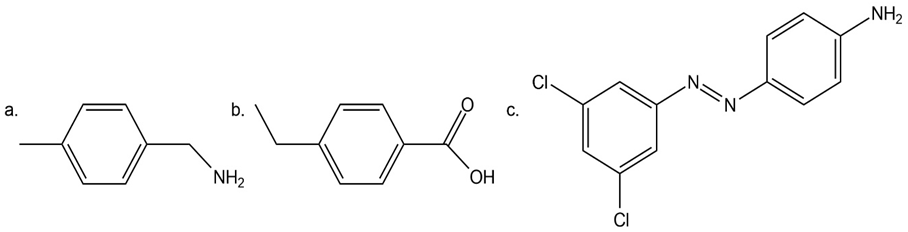
Synthesize each compound from benzene. Use a diazonium salt as one of the synthetic intermediates.
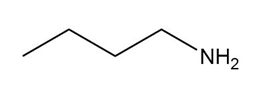
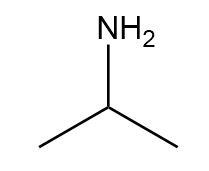
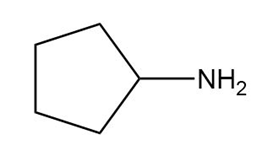
Question: Name each amine.
a.
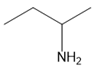
b.
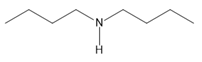
c.
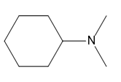
d.
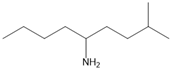
e.
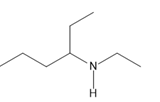
f.
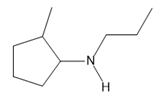
What do you think about this solution?
We value your feedback to improve our textbook solutions.