Chapter 25: PROBLEM 25.1 (page 999)
Question: Label the stereogenic centers in each compound.
a.
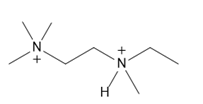
b.
Short Answer
Answer
a.
b. Compound a with a circled chiral center
Compound a with a circled chiral center
 Compound b with chiral atom circled
Compound b with chiral atom circled
Learning Materials
Features
Discover
Chapter 25: PROBLEM 25.1 (page 999)
Question: Label the stereogenic centers in each compound.
a.

b.

Answer
a.
b. Compound a with a circled chiral center
Compound a with a circled chiral center
 Compound b with chiral atom circled
Compound b with chiral atom circled
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products formed when p-methylaniline ( ) is treated with each reagent.
Explain why m-nitroaniline is a stronger base than p-nitroaniline
Which amines cannot be prepared by reduction of an amide?
a.
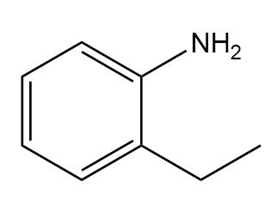
b.
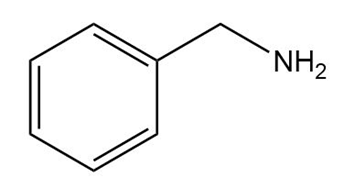
c.
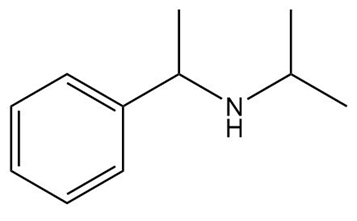
d.
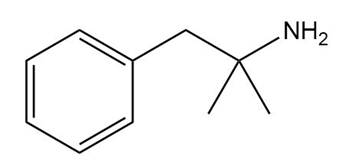
Question: What starting materials are needed to prepare each drug using reductive amination? Give all possible pairs of compounds when more than one route is possible.
a.
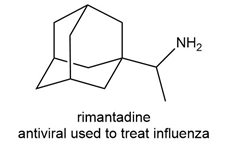
b.
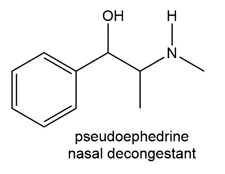
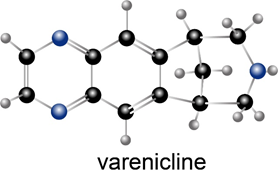
Question: Varenicline (trade name Chantix) is a drug used to help smokers quit their habit.
(a) Which N atom in varenicline is most basic? Explain your choice.
(b) What product is formed when varenicline is treated with HCl?
What do you think about this solution?
We value your feedback to improve our textbook solutions.