Chapter 25: 51 (page 1043)
How would you separate toluene ( ), benzoic acid ( ), and aniline ( ) by an extraction procedure?
Short Answer
The mixture of benzoic acid, aniline, and aniline can be extracted with the help of water and dichloromethane.
Learning Materials
Features
Discover
Chapter 25: 51 (page 1043)
How would you separate toluene ( ), benzoic acid ( ), and aniline ( ) by an extraction procedure?
The mixture of benzoic acid, aniline, and aniline can be extracted with the help of water and dichloromethane.
All the tools & learning materials you need for study success - in one app.
Get started for free
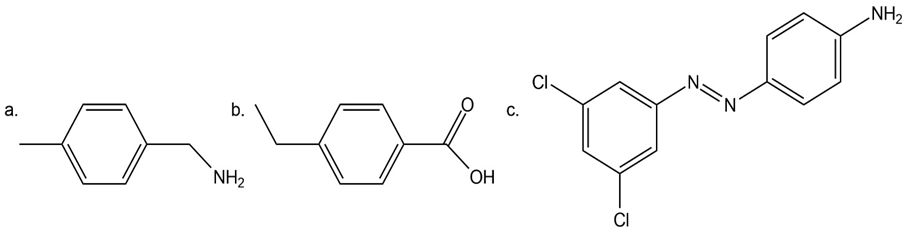
Question: Devise a synthesis of each compound from benzene. You may use any other organic or inorganic reagents.
a.
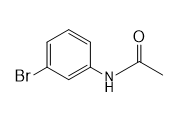
b.
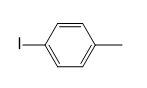
c.
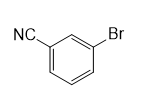
d.
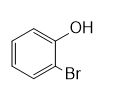
e.
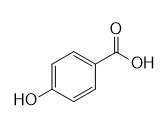
f.
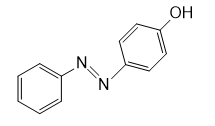
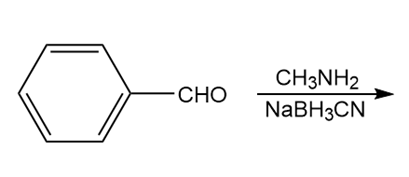
Question: Draw the product of each reaction.
a.
b.
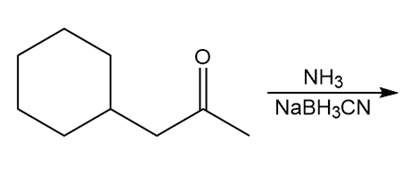
c.
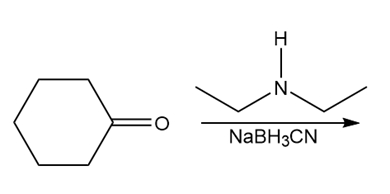
d.
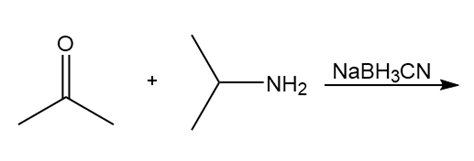
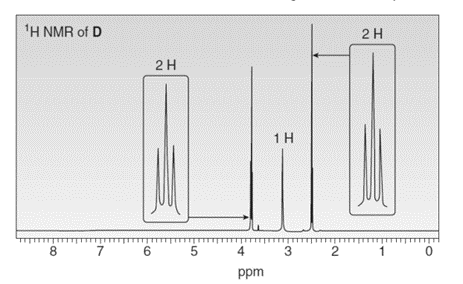
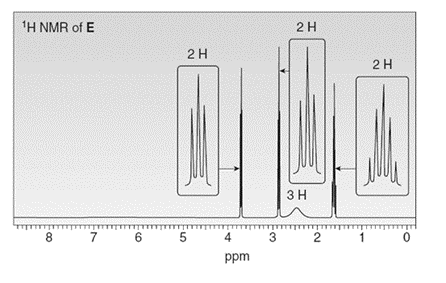
Treatment of compound D with followed by forms compound E. D shows a molecular ion in its mass spectrum at m/z= 71 and IR absorptions at and . E shows a molecular ion in its mass spectrum at m/z= 75 and IR absorptions at and . Propose structures for D and E from these data and the given 1H NMR spectra.
Question: Devise a synthesis of each compound from the given starting material(s). Albuterol is a bronchodilator and proparacaine is a local anesthetic.
Synthesize each compound from benzene. Use a diazonium salt as one of the synthetic intermediates.
What do you think about this solution?
We value your feedback to improve our textbook solutions.