Chapter 11: Q52. (page 452)
Question: Devise a synthesis of each compound using CH3CH2CH=CH2as the starting material. You may use any other compounds or inorganic reagents.
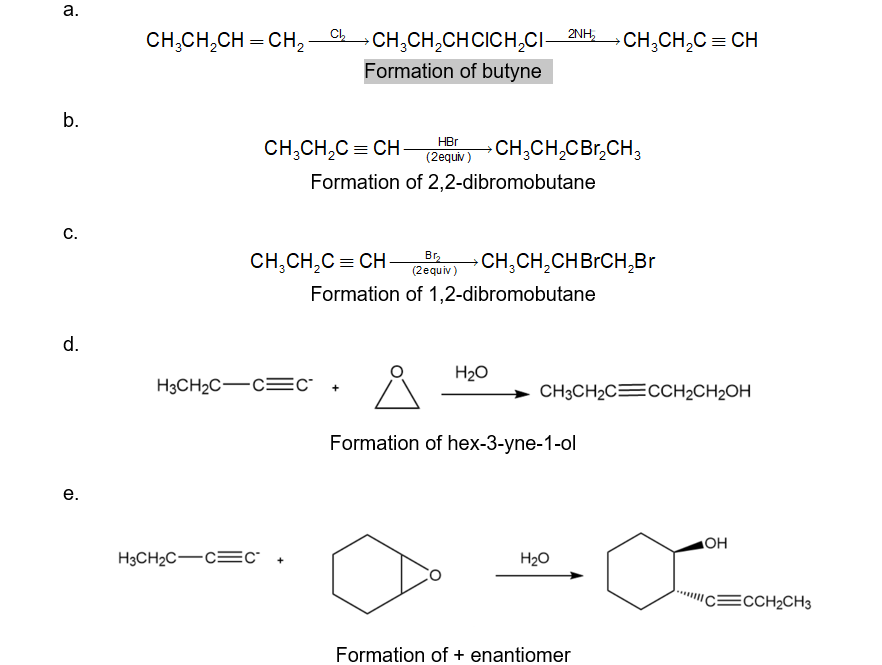
a.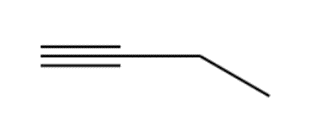
b.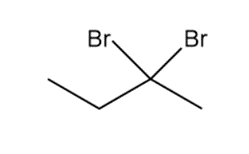
c. 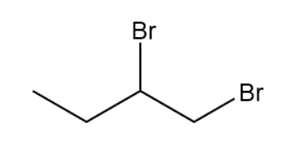
d. 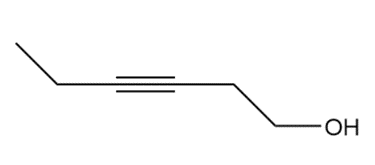
e. 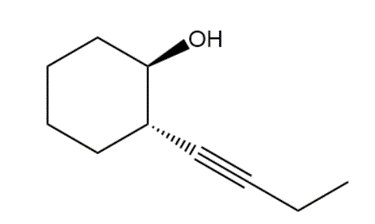
(+ enantiomer)
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 11: Q52. (page 452)
Question: Devise a synthesis of each compound using CH3CH2CH=CH2as the starting material. You may use any other compounds or inorganic reagents.
a.
b.
c. 
d. 
e. 
(+ enantiomer)
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
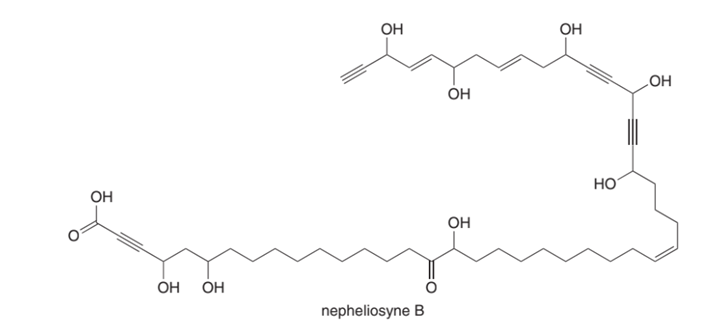
Nepheliosyne B is a novel acetylenic fatty acid isolated from a New Caledonian marine sponge.
(a) Label the most acidic H atom.
(b) Which carbon-carbon σ bond is shortest?
(c) How many degrees of unsaturation does nepheliosyne B contain?
(d) How many bonds are formed from ?
(e) Label each triple bond as internal or terminal.
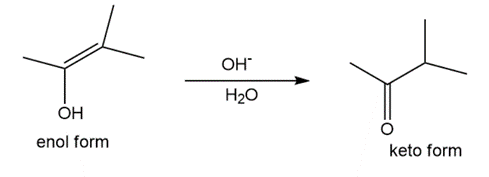
Question: Tautomerization in base resembles tautomerization in acid, but deprotonation precedes protonation in the two-step mechanism. (a) Draw a stepwise mechanism for the following tautomerization. (b) Then, draw a stepwise mechanism for the reverse reaction, the conversion of the keto form to the enol.
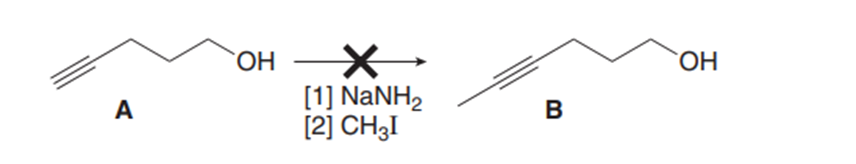
Question: When alkyne a is treated with followed by , a product having the molecular formula is formed, but it is not compound B. What is the structure of the product and why is it formed?
Give the IUPAC name for each compound.
a. 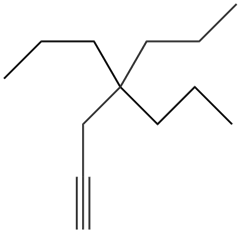
b.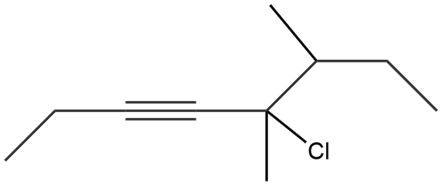
c. 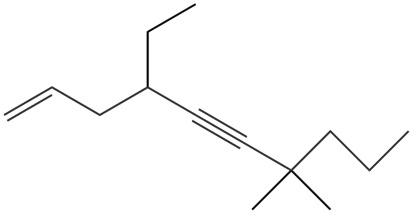
d.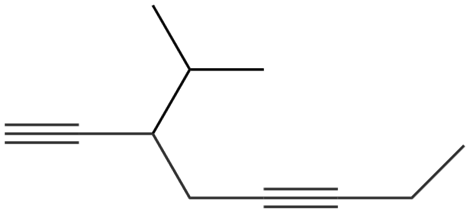
Question: Explain why an optically active solution of methylbutyrophenone loses its optical activity when dilute acid is added to the solution.
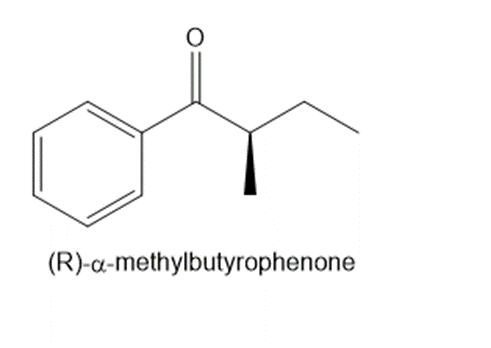
What do you think about this solution?
We value your feedback to improve our textbook solutions.