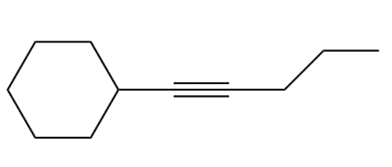
Chapter 11: Alkynes
Q41.
Question: Draw the products formed in each reaction and indicate stereochemistry.
a.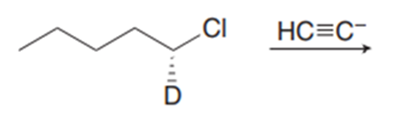
b. 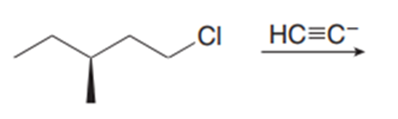
c. 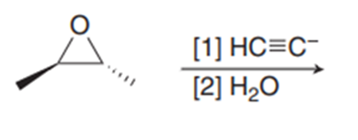
d. 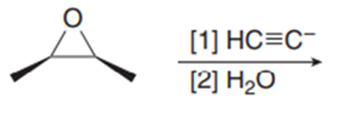
Q42.
Question: What reactions are needed to convert alcohol A into either alkyne B or alkyne C?
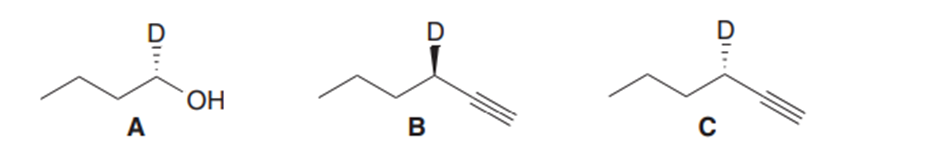
Q43.
Question: Identify the lettered compounds in the following reaction schemes. Each reaction sequence was used in the synthesis of a natural product.
a. 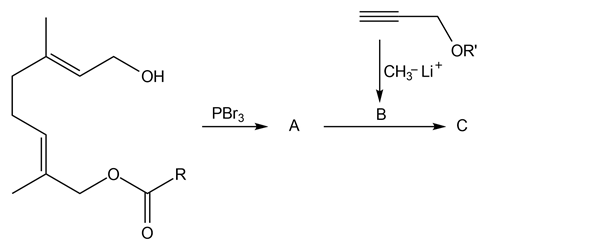
b.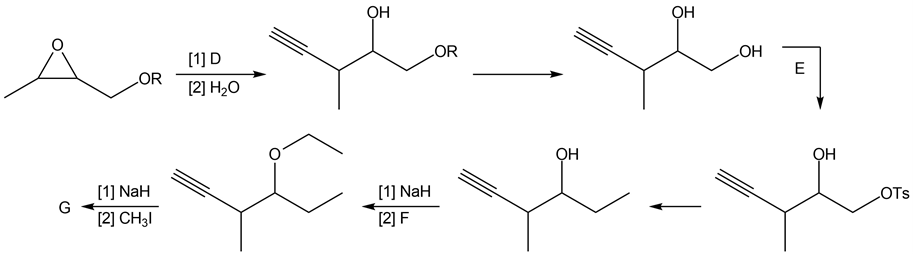
Q44.
Question: Treatment of 2,2-dibromobutane with two equivalents of strong base affords but-1-yne and but-2-yne, as well as a small amount of buta-1,2-diene. Draw a mechanism showing how each compound is formed. Which alkyne should be the major product?
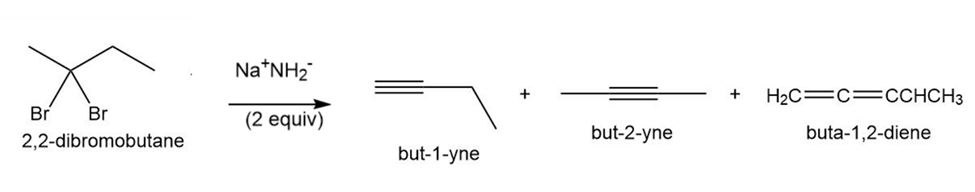
Q45.
Question: Explain the following statement. Although is more stable than,is less stable than .
Q46.
Question: Tautomerization in base resembles tautomerization in acid, but deprotonation precedes protonation in the two-step mechanism. (a) Draw a stepwise mechanism for the following tautomerization. (b) Then, draw a stepwise mechanism for the reverse reaction, the conversion of the keto form to the enol.
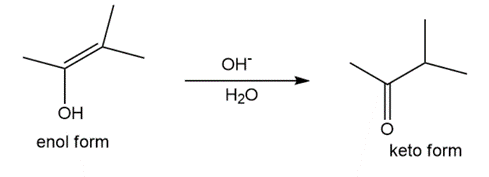
Q47.
Question: Draw a stepwise mechanism for each reaction.
a. 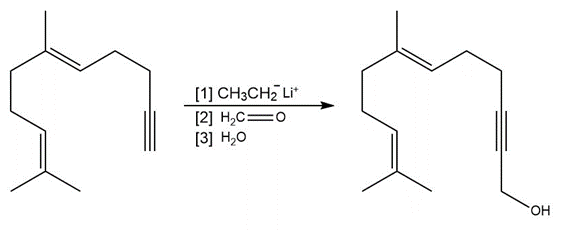
b.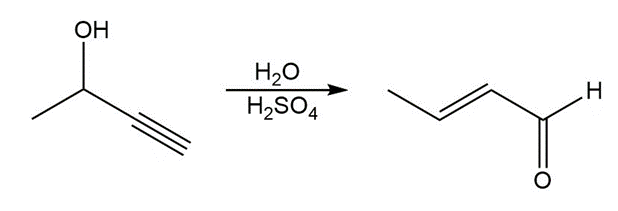
Q48.
Question: From what you have learned about enols and the hydration of alkynes, predict what product is formed by the acid-catalyzed hydration of . Draw a stepwise mechanism that illustrates how it is formed.
Q49.
Question: What steps are needed to prepare phenylacetylene, , from each compound:
Q50.
Question: The alkyl halides must be methyl or
a.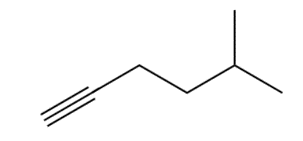
b.
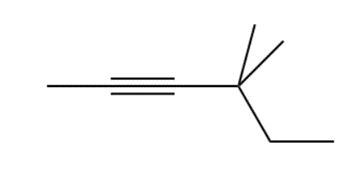
c.