Chapter 7: PROBLEM7.64 (page 293)
Question: Fluticasone, the chapter-opening molecule, can be prepared by the following reaction. Draw a stepwise mechanism for this reaction.
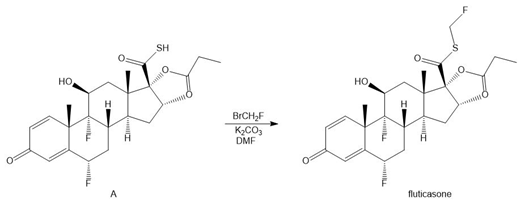
Short Answer
ANSWER
 Formation of the fluticasone by theSN1 mechanism
Formation of the fluticasone by theSN1 mechanism
Learning Materials
Features
Discover
Chapter 7: PROBLEM7.64 (page 293)
Question: Fluticasone, the chapter-opening molecule, can be prepared by the following reaction. Draw a stepwise mechanism for this reaction.

ANSWER
 Formation of the fluticasone by theSN1 mechanism
Formation of the fluticasone by theSN1 mechanism
All the tools & learning materials you need for study success - in one app.
Get started for free
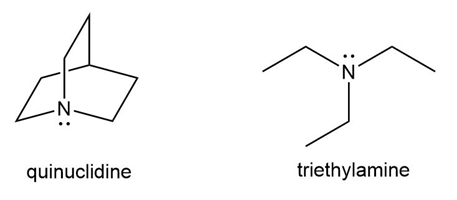
Question: Explain why quinuclidine is a much more reactive nucleophile than triethylamine, even though both compounds have N atoms surrounded by three R groups.
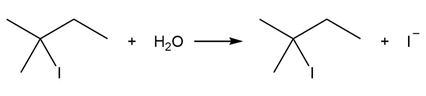
Question: Consider the following SN1 reaction.
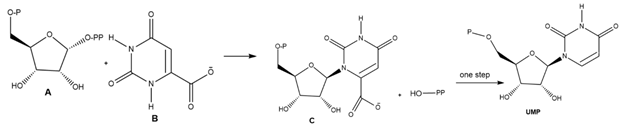
Question: Uridine monophosphate (UMP) is one of the four nucleotides that compose RNA, the nucleic acid that translates the genetic information of DNA into proteins needed by cells for proper function and development. A key step in the synthesis of UMP is the reaction of A with B to form C, which is then converted to UMP in one step. Draw a stepwise mechanism for this SN1 reaction.
Question: Device a synthesis of each compound from an alkyl halide using any other organic or inorganic reagents.
a.
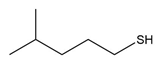
b.
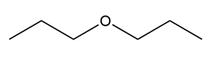
c.
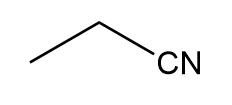
d.
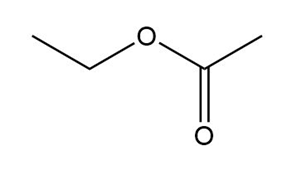
Question: When (R)-6-bromo-2,6-dimethylnonane is dissolved in , nucleophilic substitution yields an optically inactive solution. When the isomeric halide (R)-2-bromo-2,5- dimethylnonane is dissolved in under the same conditions, nucleophilic substitution forms an optically active solution. Draw the products formed in each reaction, and explain why the difference in optical activity is observed.
What do you think about this solution?
We value your feedback to improve our textbook solutions.