Chapter 7: PROBLEM 7.60 (page 292)
Question: Draw the products of each SN1 reaction and indicate the stereochemistry when necessary.
a.
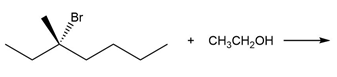
b.
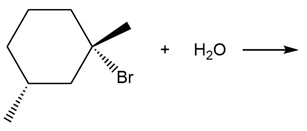
Short Answer
a.
 Products obtained from reaction (a)
Products obtained from reaction (a)
b.
 Products obtained from reaction (b)
Products obtained from reaction (b)
Learning Materials
Features
Discover
Chapter 7: PROBLEM 7.60 (page 292)
Question: Draw the products of each SN1 reaction and indicate the stereochemistry when necessary.
a.

b.

a.
 Products obtained from reaction (a)
Products obtained from reaction (a)
b.
 Products obtained from reaction (b)
Products obtained from reaction (b)
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What happens to the rate of an SN1 reaction under each of the following conditions?
a. [RX] is tripled, and stays the same.
b. Both [RX] and are tripled.
c. [RX] is halved, and stays the same.
d. [RX] is halved, andis doubled.
Question: An sp3 hybridized C-Cl bond is more polar than a sp2 hybridized C-Cl bond. (a) Explain why this phenomenon arises. (b) Rank the following compounds in order of increasing boiling point.
Question: When (R)-6-bromo-2,6-dimethylnonane is dissolved in , nucleophilic substitution yields an optically inactive solution. When the isomeric halide (R)-2-bromo-2,5- dimethylnonane is dissolved in under the same conditions, nucleophilic substitution forms an optically active solution. Draw the products formed in each reaction, and explain why the difference in optical activity is observed.
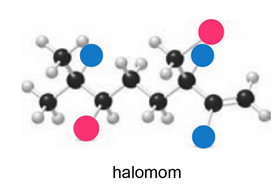
Telfairine, a naturally occurring insecticide, and halomon, an antitumor agent, are two polyhalogenated compounds isolated from red algae. (a) Classify each halide bonded to an sp3 hybridized carbon as 1°, 2°, or 3°. (b) Label each halide as vinyl, allylic, or neither.

Question: Determine the mechanism and draw the products of each reaction. Include the stereochemistry at all stereogenic centers.
a.
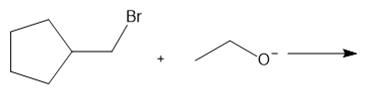
b.
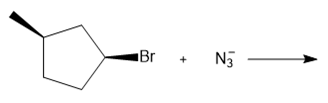
c.
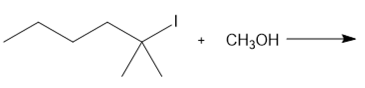
d.
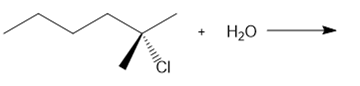
What do you think about this solution?
We value your feedback to improve our textbook solutions.