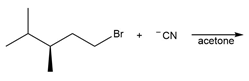
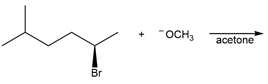
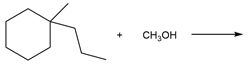
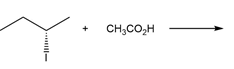
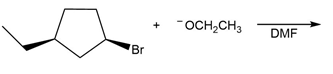Chapter 7: PROBLEM 7.39 (page 288)
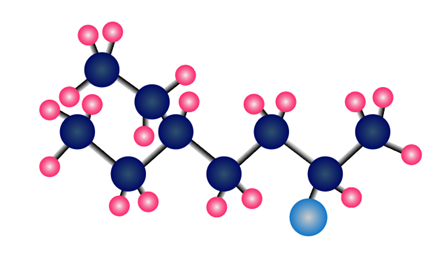
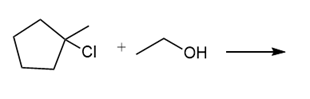
Question: The ether CH3OCH2CH3can be prepared by two different nucleophilic substitution reactions, one using CH3O- as a nucleophile and the other using CH3CH2O- as a nucleophile. Draw both routes
Short Answer
ANSWER
 Route when is used as a nucleophile
Route when is used as a nucleophile
 Route when is used as a nucleophile
Route when is used as a nucleophile
 Route when is used as a nucleophile
Route when is used as a nucleophile Route when is used as a nucleophile
Route when is used as a nucleophile