Chapter 8: Q.56 (page 329)
Draw all products, including stereoisomers, in each reaction.
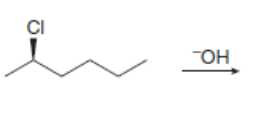
a.
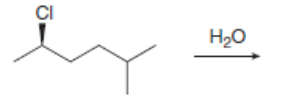
b.
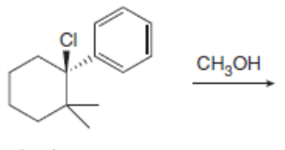
c.
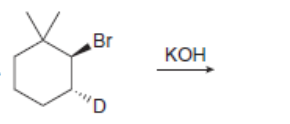
d.
Short Answer
The products formed, including their stereoisomers, are as follows:
Learning Materials
Features
Discover
Chapter 8: Q.56 (page 329)
Draw all products, including stereoisomers, in each reaction.

a.

b.

c.
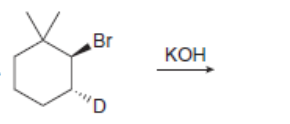
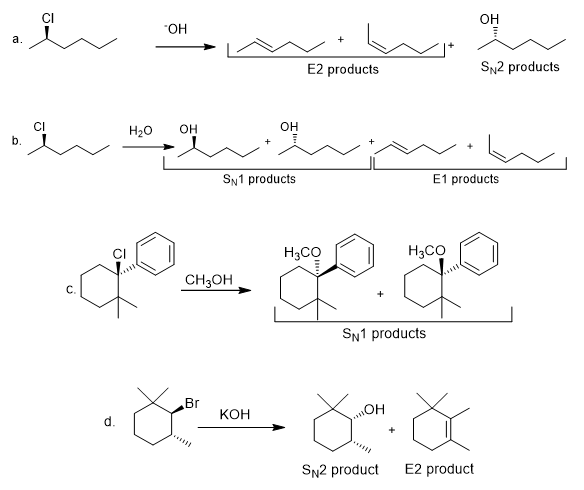
d.
The products formed, including their stereoisomers, are as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure of a dihalide that could be used to prepare each alkyne. There may be more than one possible dihalide.

a.
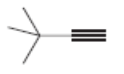
b.
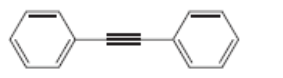
c.
For which double bonds are stereoisomers possible?
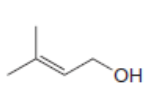
a.
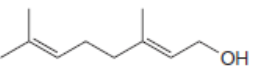
b.
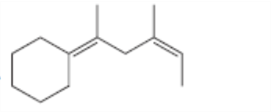
c.
Elimination of HBr from 2-bromobutane affords a mixture of but-1-ene and but-2-ene. With sodium ethoxide as base, but-2-ene constitutes 81% of the alkene products, but with potassium tert-butoxide, but-2-ene constitutes only 67% of the alkene products. Offer an explanation for this difference.
Explain why compound A does not undergo an E2 elimination with a strong base.
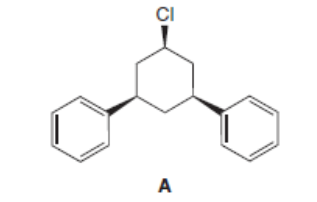
Although there are nine stereoisomers of 1,2,3,4,5,6-hexachlorocyclohexane, one stereoisomer reacts 7000 times more slowly than any of the others in an E2 elimination. Draw the structure of this isomer and explain why this is so.
What do you think about this solution?
We value your feedback to improve our textbook solutions.