Chapter 10: Q.20. (page 404)
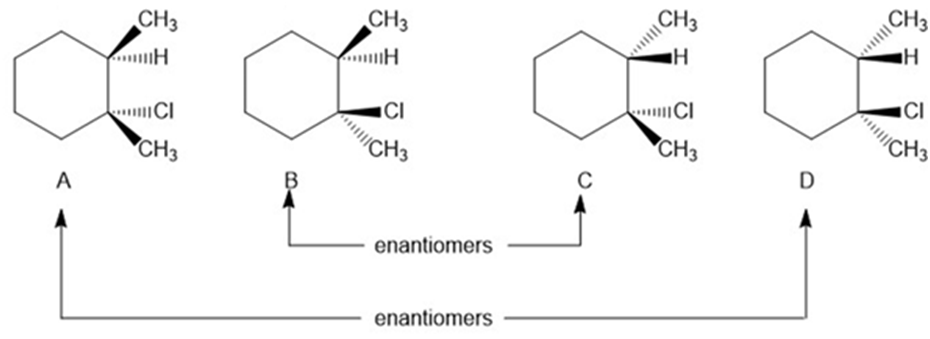
Question: Draw all stereoisomers formed when 1,2-dimethylcyclohexene is treated with HCl. Label pairs of enantiomers.
Short Answer
Answer
All the stereoisomers are shown hereunder.
Learning Materials
Features
Discover
Chapter 10: Q.20. (page 404)
Question: Draw all stereoisomers formed when 1,2-dimethylcyclohexene is treated with HCl. Label pairs of enantiomers.
Answer
All the stereoisomers are shown hereunder.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: When buta-1,3-diene (CH2=CH-CH=CH2) is treated with HBr, two constitutional isomers are formed CH3CHBr-CH=CH2and Br-CH2CH=CHCH2. Draw a stepwise mechanism that accounts for the formation of both products.
Question: Draw the products formed when (CH3)2C=CH2 is treated with each reagent.
a. HBr
b.H2O, H2SO4
c. CH3CH2HO,H2SO4
d. Cl2
e.Br2 ,H2O
f. NBS (aqueous DMSO)
g. [1] BH3; [2] H2O2 ,HO-
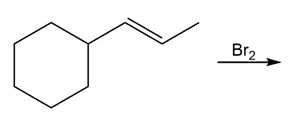
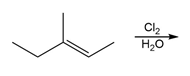
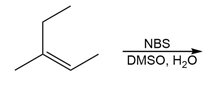
Question: Draw all stereoisomers formed in each reaction.
a.
b.
c.
Question: (a) Draw the structure of (1E,4R)-1,4-dimethylcyclodecene. (b) Draw the enantiomer and name it, including its E,Z and R,S prefixes. (c) Draw two diastereomers and name them, including the E,Z and R,S prefixes.
Question: Draw the isomers for the following compounds and then name each one:
a. 2-methyl-2,4-hexadiene
b. 2,4-heptadiene
c. 1,3-pentadiene
What do you think about this solution?
We value your feedback to improve our textbook solutions.