Chapter 21: Q9. (page 824)
Question: Which carbonyl group in each pair absorbs at a higher frequency?
a.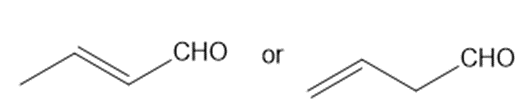
b.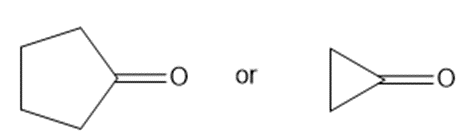
Short Answer
Answer
a. (E)-but-2-enal absorbs at a higher frequency.
b. Cyclopropanone absorbs at a higher frequency
Learning Materials
Features
Discover
Chapter 21: Q9. (page 824)
Question: Which carbonyl group in each pair absorbs at a higher frequency?
a.
b.
Answer
a. (E)-but-2-enal absorbs at a higher frequency.
b. Cyclopropanone absorbs at a higher frequency
All the tools & learning materials you need for study success - in one app.
Get started for free
What reagents are needed to convert each compound into butanal (\({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{CHO}}\))?
a.
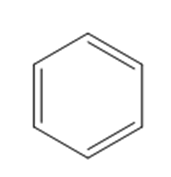
b.
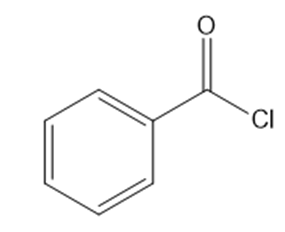
c.
Question: Give the structure corresponding to each name:
(a) sec-butyl ethyl ketone
(b) methyl vinyl ketone
(c) p-ethylacetophenone
(d) 3-benzoyl-2-benzylcyclopentanone
(e) 6,6-dimethylcyclohex-2-enone
(f) 3-ethylhex-5-enal
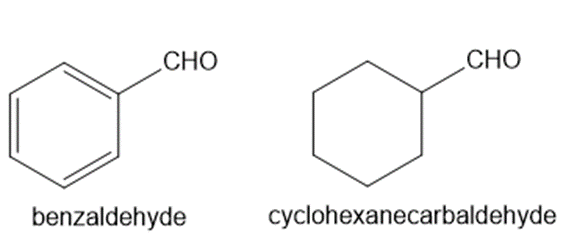
Question: Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards nucleophilic attack.
Question: The boiling point of butan-2-one ( 80 °C ) is significantly higher than the boiling point of diethyl ether (35 °C ), even though both compounds exhibit dipole–dipole interactions and have comparable molecular weights. Offer an explanation
Question: Give the IUPAC name for each aldehyde.
4. 3,6-diethylnonanal
What do you think about this solution?
We value your feedback to improve our textbook solutions.