Chapter 9: Q52. (page 377)
Question: Draw a stepwise mechanism for the following reaction.
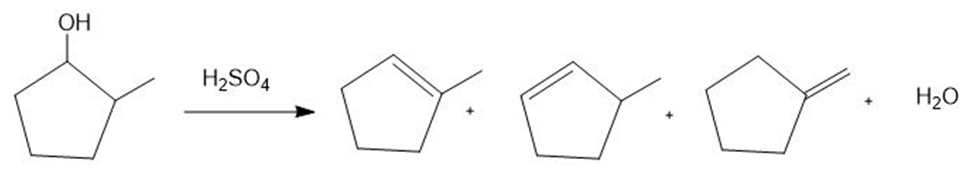
Short Answer
Answer
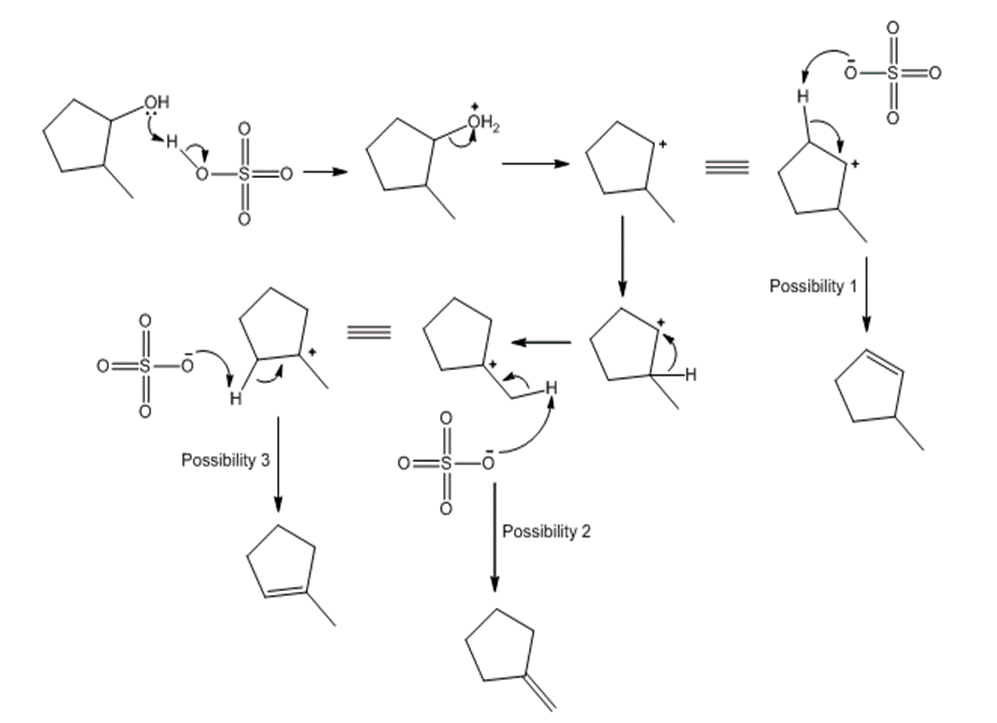
Learning Materials
Features
Discover
Chapter 9: Q52. (page 377)
Question: Draw a stepwise mechanism for the following reaction.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Show how a 1,2-shift forms a more stable carbocation from each intermediate.
a.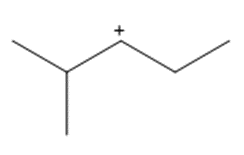
b. 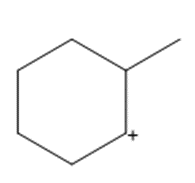
c. 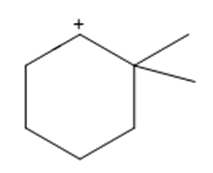
Question: Draw the products formed when (S)-butan-2-ol is treated with TsCl and pyridine, followed by NaOH. Label the stereogenic center in each compound as R or S. What is the stereochemical relationship between the starting alcohol and the final product?
Question: Give the structure corresponding to each name.
a. trans-2-methylcyclohexanol
b. 2, 3, 3-trimethylbutan-2-ol
c. 6-sec-butyl-7, 7-diethyldecan-4-ol
d. 3-chloropropane-1, 2-diol
e. 1, 2-epoxy-1, 3, 3-trimethylcyclohexane
f. 1-ethoxy-3-ethylheptane
g. (2R,3S)-3-isopropylhexan-2-ol
h. (S)-2-ethoxy-1, 1-dimethylcyclopentane
i. 4-ethylheptane-3-thiol
j. 1-isopropylthio-2-methylcyclohexane
Question: When each halohydrin is treated with NaH a product of molecular formulais formed. Draw the structure of the product and indicate its stereochemistry.
a.
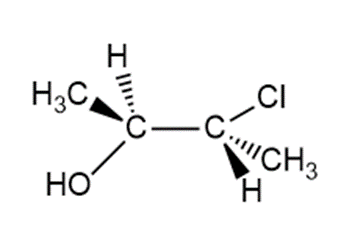
b.
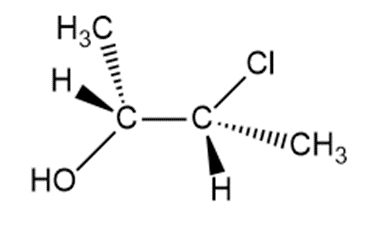
c.
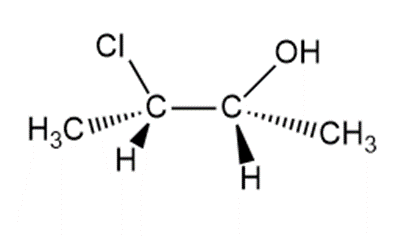
Question: Draw the products formed when ethylene oxide is treated with each reagent.
What do you think about this solution?
We value your feedback to improve our textbook solutions.