Chapter 30: Question 30.3 (page 1202)
What polymer is formed by the radical polymerization of each monomer?
(a.)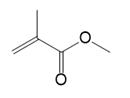
(b.)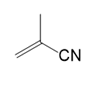
Short Answer
Answer
(a.)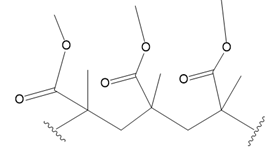
(b.)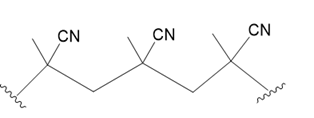
Learning Materials
Features
Discover
Chapter 30: Question 30.3 (page 1202)
What polymer is formed by the radical polymerization of each monomer?
(a.)
(b.)
Answer
(a.)
(b.)
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure of the polymer formed by step-growth polymerization of each monomer or pair of monomers.
(a.)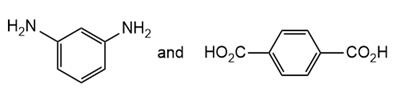
(b.)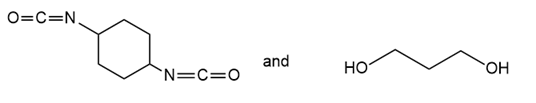
(c.)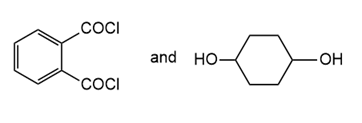
(d.)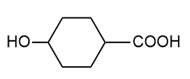
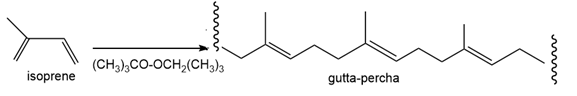
Draw a stepwise mechanism for the polymerization of isoprene to gutta-percha usingas the initiator.
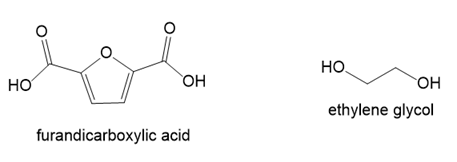
Draw the structure of PEF, polyethylene furanoate, a condensation polymer formed from furandicarboxylic acid and ethylene glycol. PEF, which can be synthesized from precursors that are obtained from renewable resources, has many of the same properties as polyethylene terephthalate (PET).
Explain why acrylonitrile () undergoes cationic polymerization more slowly than but-3-enenitrile () .
(a) Draw the structure of the prepolymer A formed from 1,4-dihydroxybenzene and excess epichlorohydrin. (b) Draw the structure of the cross-linked polymer B formed when A is treated with as the hardening agent.
What do you think about this solution?
We value your feedback to improve our textbook solutions.