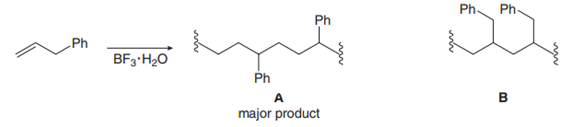
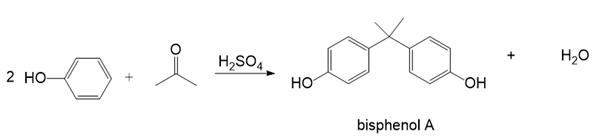
Chapter 30: Question 30.16 (page 1213)
Draw the structure of PEF, polyethylene furanoate, a condensation polymer formed from furandicarboxylic acid and ethylene glycol. PEF, which can be synthesized from precursors that are obtained from renewable resources, has many of the same properties as polyethylene terephthalate (PET).
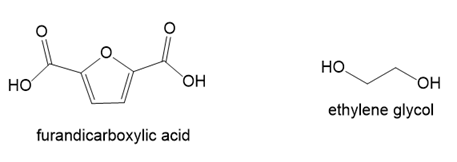
Short Answer
Answer
 Polyethylene furanoate
Polyethylene furanoate
 Formation of polyethylene furanoate
Formation of polyethylene furanoate

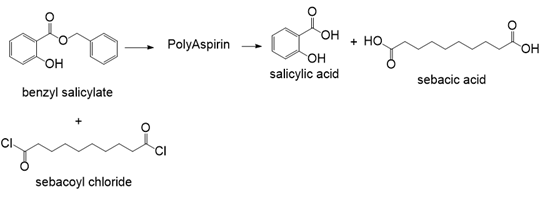
 Poly(ester amide) A
Poly(ester amide) A