Chapter 30: Question 30.29 (page 1225)
What monomer(s) are used to prepare each polymer or copolymer?
(a.)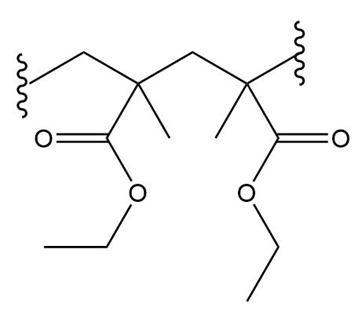
(b.)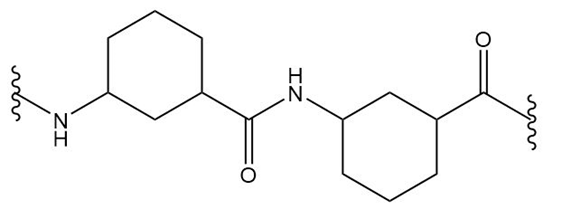
(c.)
(d.)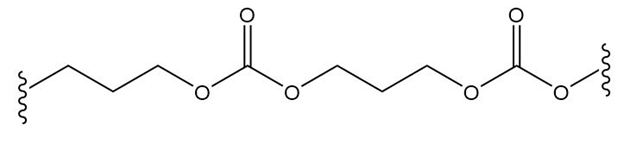
Short Answer
Answer
(a.)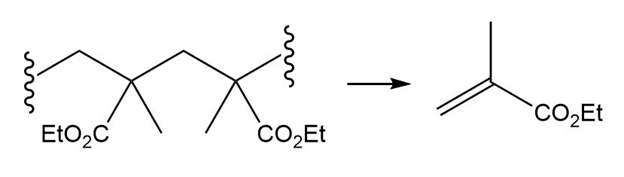
(b.)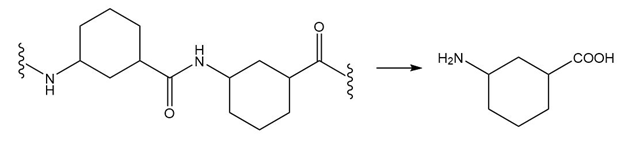
(c.)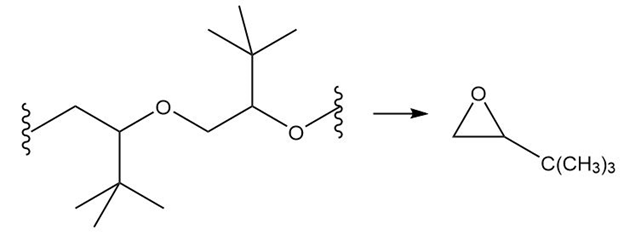
(d.)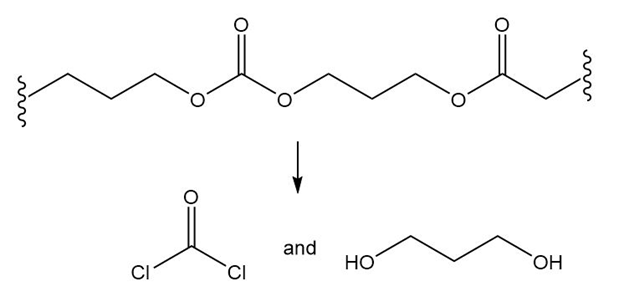
Learning Materials
Features
Discover
Chapter 30: Question 30.29 (page 1225)
What monomer(s) are used to prepare each polymer or copolymer?
(a.)
(b.)
(c.)
(d.)
Answer
(a.)
(b.)
(c.)
(d.)
All the tools & learning materials you need for study success - in one app.
Get started for free
The polymerization under radical conditions affords products A and B. Draw a mechanism that accounts for their formation.
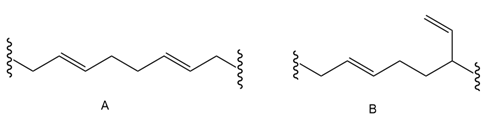
Draw a short segment of each polymer:
(a) Isotactic poly(vinyl chloride);
(b) Syndiotactic polyacrylonitrile;
(c) Atactic polystyrene.
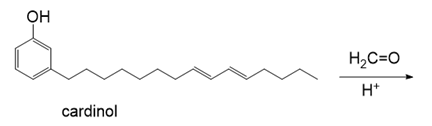
Thermosetting resins similar to Bakelite (Section 30.7) have also been prepared from renewable feedstocks. One method uses cardinol, the major constituent of the liquid obtained from roasted cashew nutshells. What polymer is obtained when cardinol is treated with formaldehyde () in the presence of a proton source?
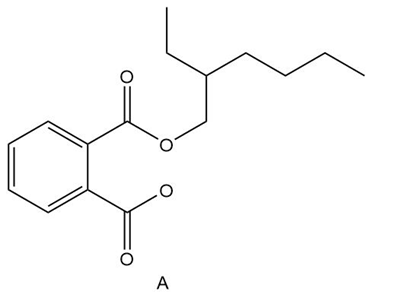
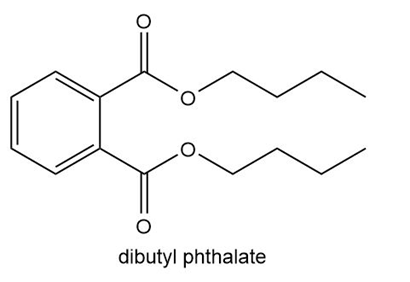
Explain why diester A is now often used as a plasticizer in place of dibutyl phthalate.
Explain why cationic polymerization is an effective method of polymerizing but not .
What do you think about this solution?
We value your feedback to improve our textbook solutions.