Chapter 30: Question 30.14 (page 1211)
The polymerization under radical conditions affords products A and B. Draw a mechanism that accounts for their formation.
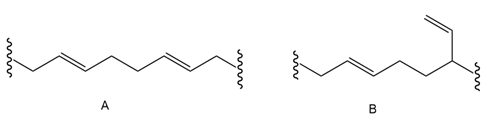
Short Answer
Answer
 Mechanism of the reaction
Mechanism of the reaction
Learning Materials
Features
Discover
Chapter 30: Question 30.14 (page 1211)
The polymerization under radical conditions affords products A and B. Draw a mechanism that accounts for their formation.

Answer
 Mechanism of the reaction
Mechanism of the reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the products of each reaction.
(a.)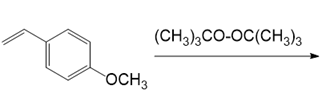
(b.)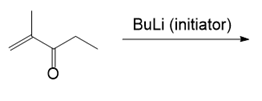
(c.)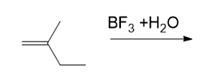
(d.)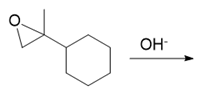
(e.)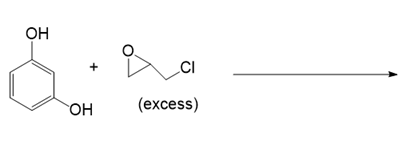
(f.)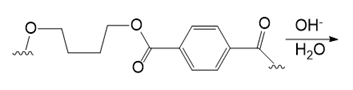
(g.)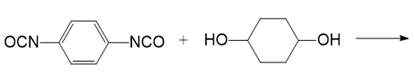
(h.)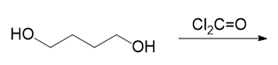
Draw the alternating copolymer formed from each set of monomers.
(a.) and
and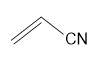
(b.)
 and
and
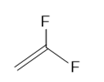
Draw a stepwise mechanism for the anionic polymerization of styrene to form polystyrene using BuLi as an initiator. Use CO2 as the electrophile that terminates the chain mechanism.
Draw a stepwise mechanism for the conversion of acrylonitrile to polyacrylonitrile, , using butyllithium(BuLi) as the initiator and CO2 as the electrophile to terminate the chain.
Draw the structures of Quiana and Nomex, two commercially available step-growth polymers formed from the given monomers. Nomex is a strong polymer used in aircraft tires and microwave transformers. Quiana has been used to make wrinkle-resistant fabrics.
(a.)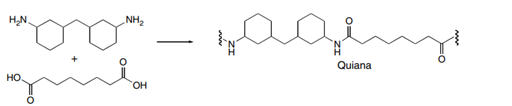
(b.)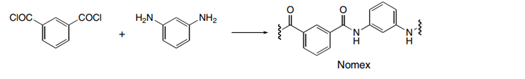
What do you think about this solution?
We value your feedback to improve our textbook solutions.