Chapter 30: Question 30.8 (page 1206)
Draw a stepwise mechanism for the conversion of acrylonitrile to polyacrylonitrile, , using butyllithium(BuLi) as the initiator and CO2 as the electrophile to terminate the chain.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 30: Question 30.8 (page 1206)
Draw a stepwise mechanism for the conversion of acrylonitrile to polyacrylonitrile, , using butyllithium(BuLi) as the initiator and CO2 as the electrophile to terminate the chain.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a stepwise mechanism for the anionic polymerization of styrene to form polystyrene using BuLi as an initiator. Use CO2 as the electrophile that terminates the chain mechanism.
What polymer is formed by chain-growth polymerization of each monomer?
(a.)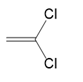
(b.)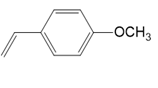
(c.)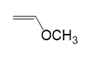
(d.)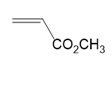
Explain why cationic polymerization is an effective method of polymerizing but not .
In addition to glycolic and lactic acids (Section 30.6B), dissolving sutures can also be prepared from each of the following lactone monomers. Draw the structure of the polymer formed from each monomer.
(a.)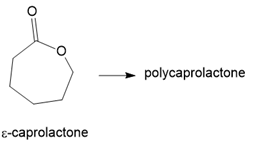
(b.)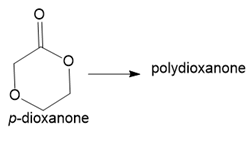
Give the shorthand structures of poly(vinyl chloride) and nylon 6,6 in Section 30.1.
What do you think about this solution?
We value your feedback to improve our textbook solutions.