Chapter 30: Question 30.2 (page 1201)
What polymer is formed by chain-growth polymerization of each monomer?
(a.)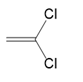
(b.)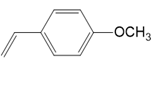
(c.)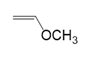
(d.)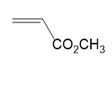
Short Answer
Answer
(a.)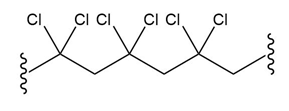
(b.)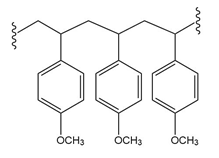
(c.)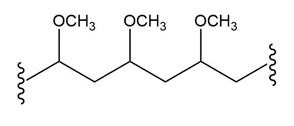
(d.)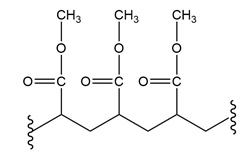
Learning Materials
Features
Discover
Chapter 30: Question 30.2 (page 1201)
What polymer is formed by chain-growth polymerization of each monomer?
(a.)
(b.)
(c.)
(d.)
Answer
(a.)
(b.)
(c.)
(d.)
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structures of Quiana and Nomex, two commercially available step-growth polymers formed from the given monomers. Nomex is a strong polymer used in aircraft tires and microwave transformers. Quiana has been used to make wrinkle-resistant fabrics.
(a.)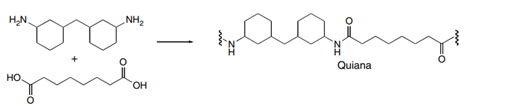
(b.)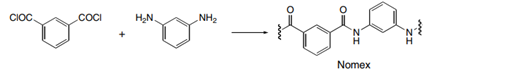
Although chain branching in radical polymerizations can occur by intermolecular H abstraction as shown in Mechanism 30.2, chain branching can also occur by intramolecular H abstraction to form branched polyethylene that contains butyl groups as branches.
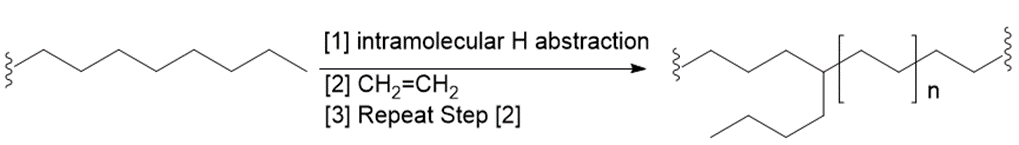
a. Draw a stepwise mechanism that illustrates which H must be intramolecularly abstracted to form butyl substituents.
b. Suggest a reason why the abstraction of this H is more facile than the abstraction of other H’s.
Give the shorthand structures of poly(vinyl chloride) and nylon 6,6 in Section 30.1.
Explain why radical polymerization of styrene forms branched chains with 4° carbons as in A, but none with 3° carbons as in B.
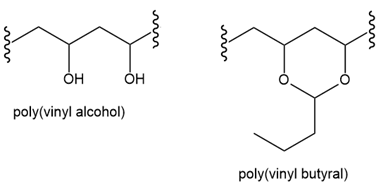
(a) Explain why poly(vinyl alcohol) cannot be prepared by the radical polymerization of vinyl alcohol (b) Devise a stepwise synthesis of poly(vinyl alcohol) from vinyl acetate(c) How can poly(vinyl alcohol) be converted to poly(vinyl butyral), a polymer used in windshield safety glass?
What do you think about this solution?
We value your feedback to improve our textbook solutions.