Chapter 30: Question 30.28 (page 1225)
Draw the structure of the alternating copolymer formed from each pair of monomers.
(a.)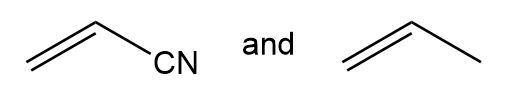
(b.)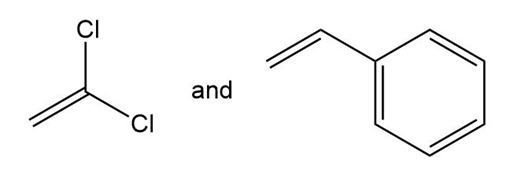
(c.)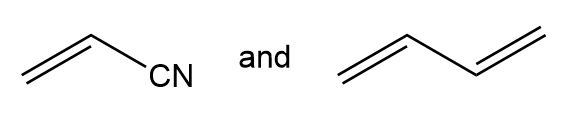
(d.)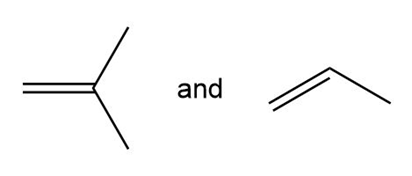
Short Answer
Answer
(a.)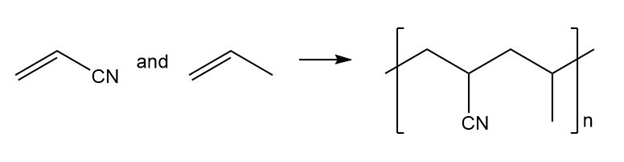
(b.)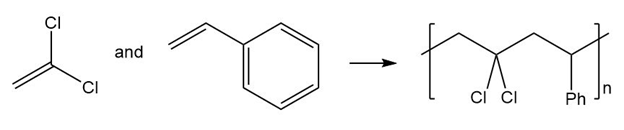
(c.)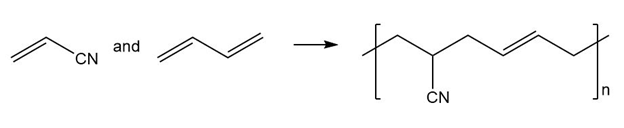
(d.)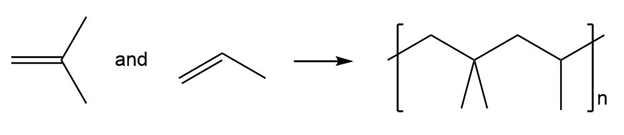
Learning Materials
Features
Discover
Chapter 30: Question 30.28 (page 1225)
Draw the structure of the alternating copolymer formed from each pair of monomers.
(a.)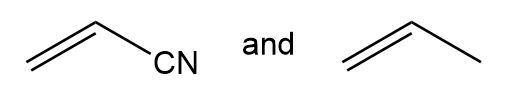
(b.)
(c.)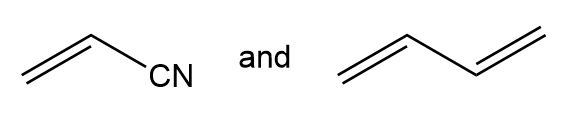
(d.)
Answer
(a.)
(b.)
(c.)
(d.)
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the mechanism for the radical polymerization of vinyl acetate using as the initiator.
Which method of ionic polymerization-cationic or anionic-is preferred for each monomer? Explain your choices.
(a.)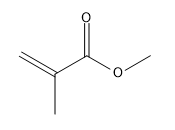
(b.)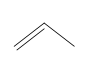
(c.)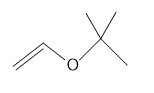
(d.)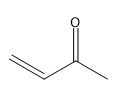
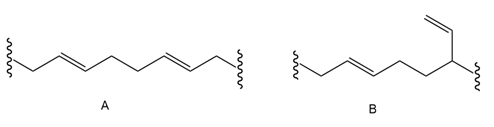
The polymerization under radical conditions affords products A and B. Draw a mechanism that accounts for their formation.
(a) Draw the structure of the prepolymer A formed from 1,4-dihydroxybenzene and excess epichlorohydrin. (b) Draw the structure of the cross-linked polymer B formed when A is treated with as the hardening agent.
Draw a stepwise mechanism for the conversion of acrylonitrile to polyacrylonitrile, , using butyllithium(BuLi) as the initiator and CO2 as the electrophile to terminate the chain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.