Chapter 30: Question 30.39 (page 1226)
Draw a stepwise mechanism for the polymerization of isoprene to gutta-percha usingas the initiator.
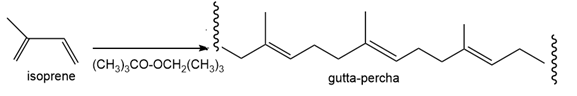
Short Answer
Answer
 Mechanism
Mechanism
Learning Materials
Features
Discover
Chapter 30: Question 30.39 (page 1226)
Draw a stepwise mechanism for the polymerization of isoprene to gutta-percha usingas the initiator.

Answer
 Mechanism
Mechanism
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure of the polymer formed by step-growth polymerization of each monomer or pair of monomers.
(a.)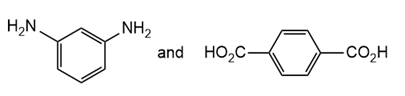
(b.)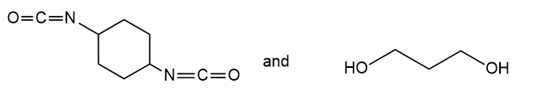
(c.)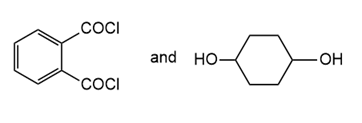
(d.)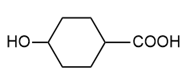
Assign Eor Zconfiguration to the double bonds in neoprene. Draw a stereoisomer of neoprene in which all the double bonds have the opposite configuration.
(a) Draw the structure of the prepolymer A formed from 1,4-dihydroxybenzene and excess epichlorohydrin. (b) Draw the structure of the cross-linked polymer B formed when A is treated with as the hardening agent.
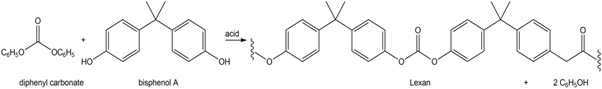
Lexan can also be prepared by the acid-catalyzed reaction of diphenyl carbonate with bisphenol A. Draw a stepwise mechanism for this process.
Explain why radical polymerization of styrene forms branched chains with 4° carbons as in A, but none with 3° carbons as in B.
What do you think about this solution?
We value your feedback to improve our textbook solutions.