Chapter 30: Question 30.18 (page 1214)
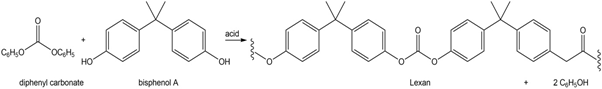
Lexan can also be prepared by the acid-catalyzed reaction of diphenyl carbonate with bisphenol A. Draw a stepwise mechanism for this process.
Short Answer
Answer
 Mechanism of the reaction
Mechanism of the reaction
Learning Materials
Features
Discover
Chapter 30: Question 30.18 (page 1214)
Lexan can also be prepared by the acid-catalyzed reaction of diphenyl carbonate with bisphenol A. Draw a stepwise mechanism for this process.

Answer
 Mechanism of the reaction
Mechanism of the reaction
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the structure of the polymer formed by step-growth polymerization of each monomer or pair of monomers.
(a.)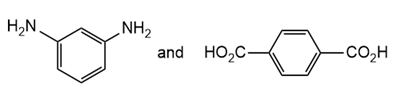
(b.)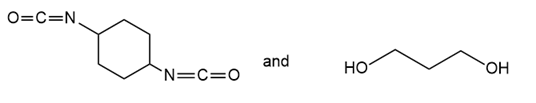
(c.)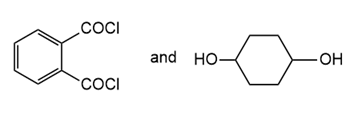
(d.)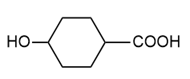
What polyamide is formed from each monomer or pair of monomers?
(a.)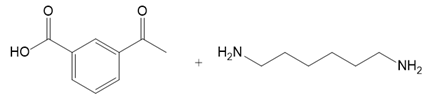
(b.)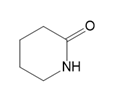
(c.)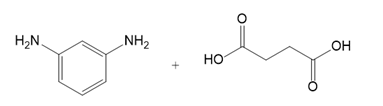
Explain why acrylonitrile () undergoes cationic polymerization more slowly than but-3-enenitrile () .
Draw a short segment of each polymer:
(a) Isotactic poly(vinyl chloride);
(b) Syndiotactic polyacrylonitrile;
(c) Atactic polystyrene.
ABS, a widely produced copolymer used in crash helmets, small appliances, and toys, is formed from three monomers- acrylonitrile , buta-1,3-diene , and styrene (CH2 =CHPh). draw a possible structure for ABS.
What do you think about this solution?
We value your feedback to improve our textbook solutions.