Chapter 30: Question 30.11 (page 1207)
ABS, a widely produced copolymer used in crash helmets, small appliances, and toys, is formed from three monomers- acrylonitrile , buta-1,3-diene , and styrene (CH2 =CHPh). draw a possible structure for ABS.
Short Answer
Answer
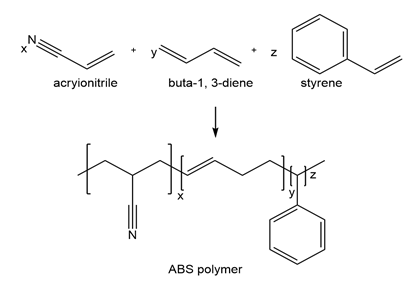
 Monomers of ABS
Monomers of ABS Structure of ABS
Structure of ABS

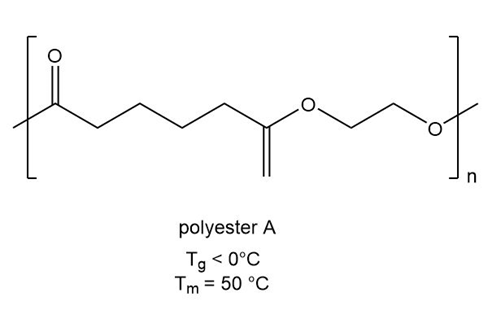
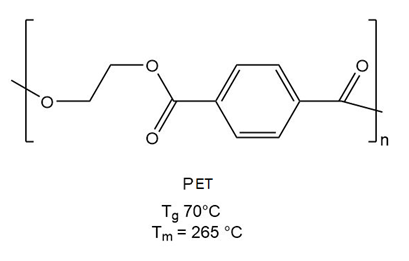
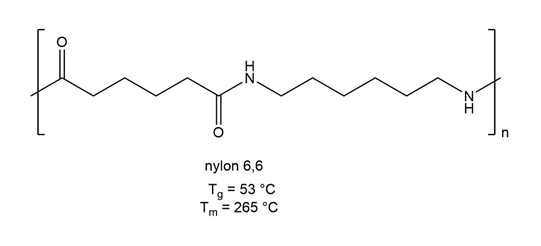
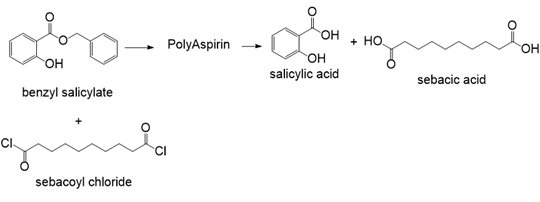
 PBAT
PBAT