Chapter 27: Pericyclic Reactions
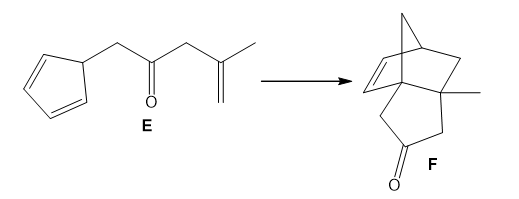
Q 56.
Question: Use curved arrows to show how E is converted to F by a two-step reaction sequence consisting of a [1,5] sigmatropic rearrangement followed by a [4 + 2] cycloaddition.
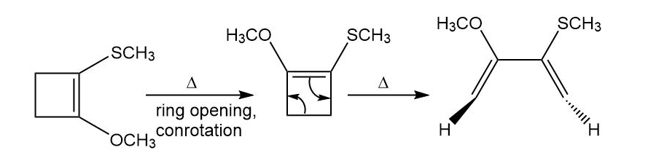
Q 57.
Question: Show how the following starting material is converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.
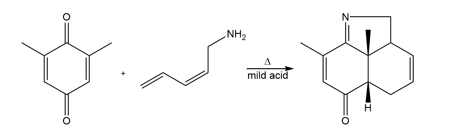
Q 58.
Question: Draw a stepwise, detailed mechanism for the following reaction.
Q 59.
Question: What product is formed by [3,3] sigmatropic rearrangement of the following compound? Clearly indicate the stereochemistry around all tetrahedral stereogenic centers.
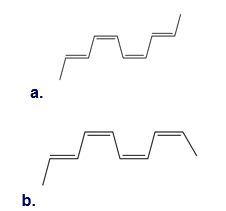
Q 6.
Question: What product is formed when each compound undergoes thermal electrocyclic ring-opening or ring closure? Label each process as conrotatory or disrotatory and clearly indicate the stereochemistry around tetrahedral stereogenic centers and double bonds.
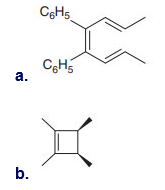
Q 60.
Question: a) What is the structure of C, which is formed by oxy-Cope rearrangement of B with NaOEt?
(b) Draw a stepwise mechanism for the conversion of C to the bicyclic alcohol D.
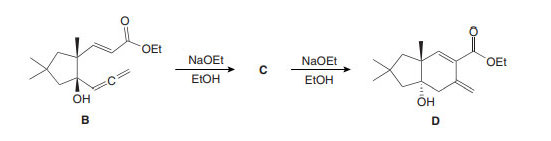
Q 61.
Question:Draw a stepwise mechanism for the Carroll rearrangement, a reaction that prepares aγ,δ-unsaturated carbonyl compound from aβ-keto ester and allylic alcohol in the presence ofbase.
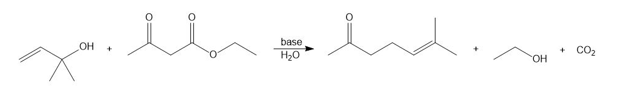
Q 62.
Question: The endiandric acids comprise a group of unsaturated carboxylic acids isolated from a tree that grows in the rain forests of eastern Australia. The methyl esters of endiandric acids D and E have been prepared from polyene Y by a series of two successive electrocyclic reactions: thermal ring closure of the conjugated tetraene followed by ring closure of the resulting conjugated triene. (a) Draw the structures (including stereochemistry) of the methyl esters of endiandric acids D and E. (b) The methyl ester of endiandric acid E undergoes an intramolecular [4 + 2] cycloaddition to form the methyl ester of endiandric acid A. Propose a possible structure for endiandric acid A.
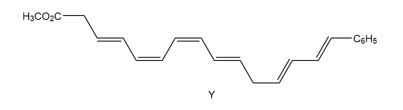
Q 63.
Question: [4 + 2] Cycloadditions with o-quinones such as B are often complex because a variety of products are possible.a. Draw arrows to illustrate how each product is formed when B reacts with styrene, and label the “diene” and “dienophile” components.
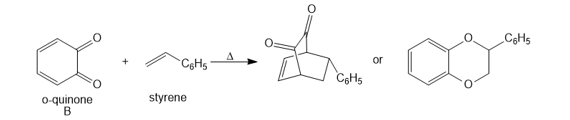
b. Draw arrows to illustrate how each product is formed when B reacts with buta-1,3-diene, and label the “diene” and “dienophile” components.
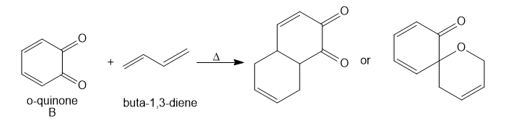
c. o-Quinone C reacts with diene D to form heterocycle E by a process that involves a cycloaddition followed by a [3,3] sigmatropic rearrangement. Use curved arrows to illustrate how this two-step sequence occurs. E is not formed directly from C by a Diels– Alder reaction.
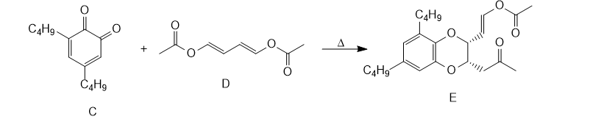
Q 7.
Question: What cyclic product is formed when each decatetraene undergoes thermal electrocyclic ring closure?