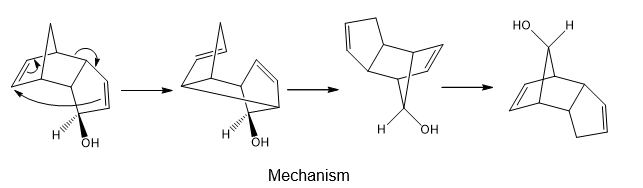
Chapter 27: Q 59. (page 1104)
Question: What product is formed by [3,3] sigmatropic rearrangement of the following compound? Clearly indicate the stereochemistry around all tetrahedral stereogenic centers.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 27: Q 59. (page 1104)
Question: What product is formed by [3,3] sigmatropic rearrangement of the following compound? Clearly indicate the stereochemistry around all tetrahedral stereogenic centers.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
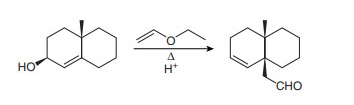
Question: Draw a stepwise, detailed mechanism for the following reaction.
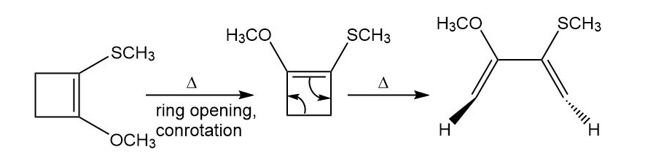
Question: Show how the following starting material is converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.
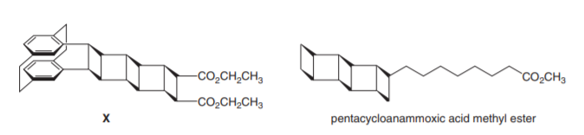
Question: How can X be prepared from a constitutional isomer by a series of cycloaddition reactions? Interest in molecules that contain several cyclobutane rings fused together has been fueled by the discovery of pentacycloanammoxic acid methyl ester, a lipid isolated from the membrane of organelles in the bacterium Candidatus Brocadia anammoxidans. The role of this unusual natural product is as yet unknown.
Question:Show that a thermal suprafacial addition is symmetry allowed in a [4 + 2] cycloaddition by using the HOMO of the alkene and the LUMO of the diene.
Question: What product is formed when each compound in Problem 27.6 undergoes photochemical electrocyclic ring-opening or ring closure? Label each process as conrotatory or disrotatory and clearly indicate the stereochemistry around tetrahedral stereogenic centers and double bonds.
What do you think about this solution?
We value your feedback to improve our textbook solutions.